Ellacor
Newport Beach
It’s Time To Reveal Your Inner Youth — With Less Compromise!








We are thrilled to announce that The Aesthetic Centers, has been recognized as an Ellacor Top 25 Procedure Provider!
Being recognized as an ellacor Top 25 Procedure Provider reaffirms our dedication to excellence in every aspect of our practice. From consultation to recovery, our team is committed to providing personalized and exceptional experiences for each patient. Your aesthetic journey is our priority, and this prestigious distinction reflects our commitment to your care.
Embark on your transformative journey with confidence, knowing that you are in the capable hands of an ellacor Top 25 Procedure Provider. Book your complimentary consult today.
The Aesthetic Centers was selected as one of the Top Plastic Surgery Offices to receive the Ellacor™ System in 2021, along with another 6 offices nationwide.
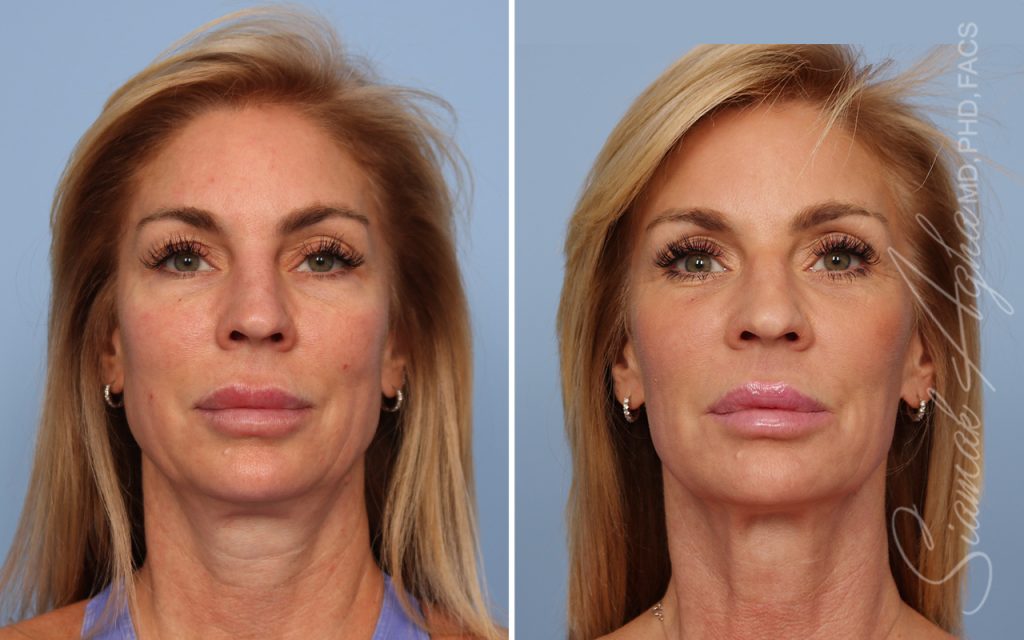
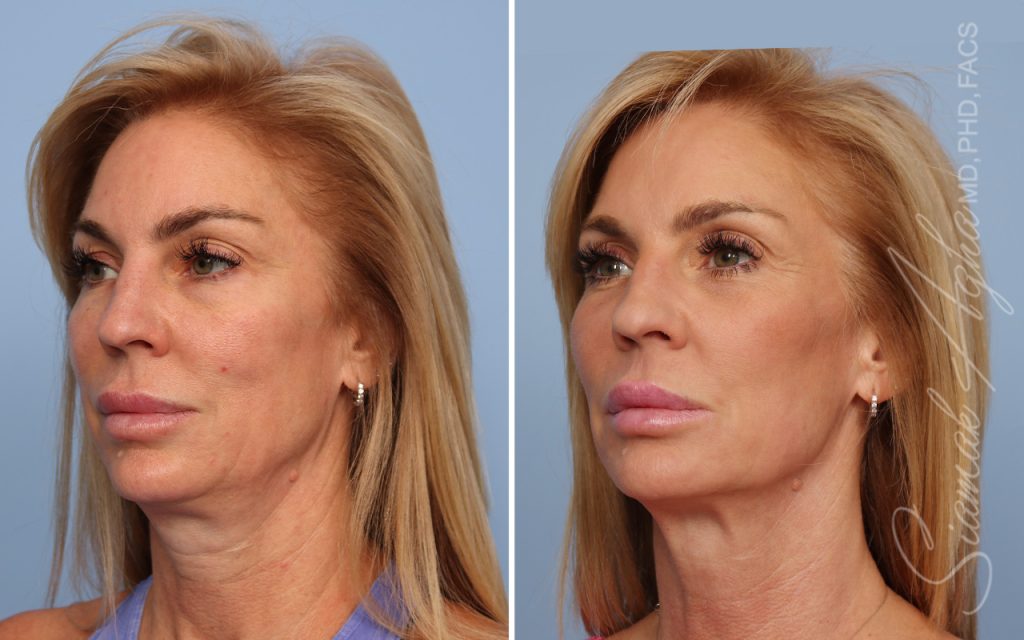
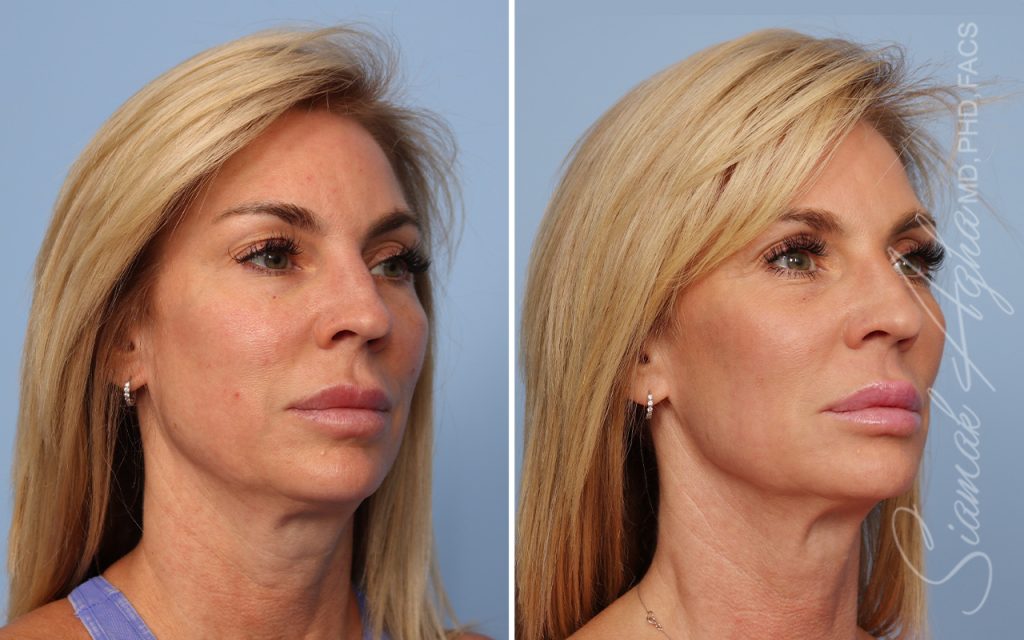
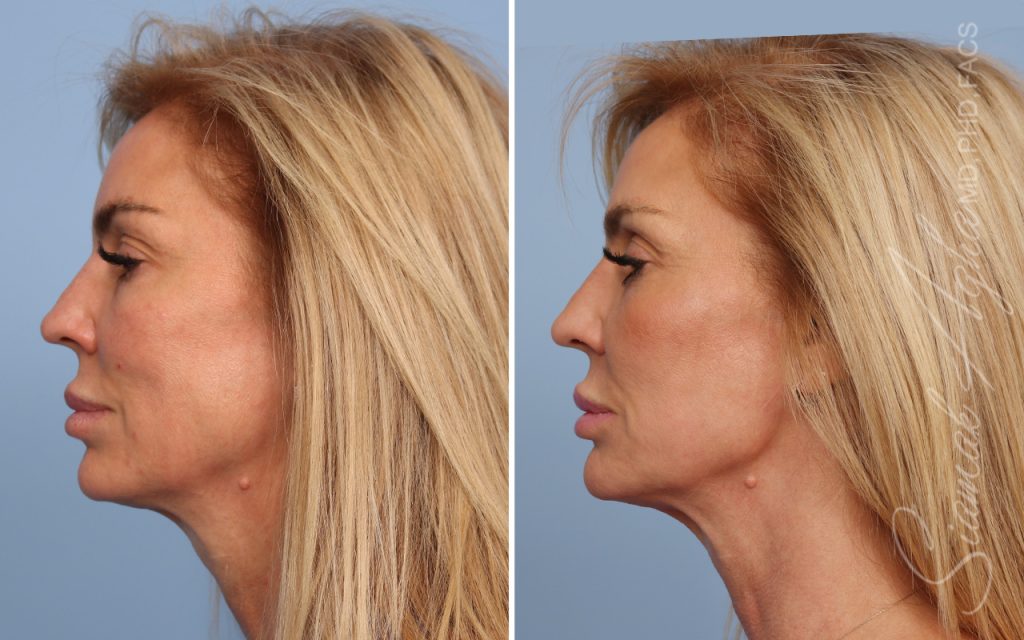
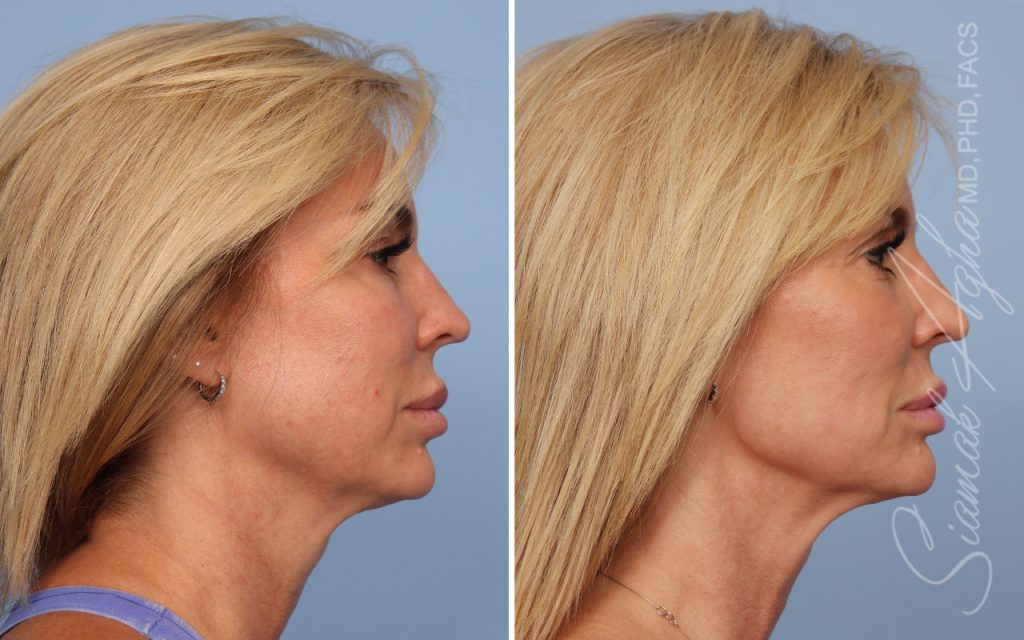
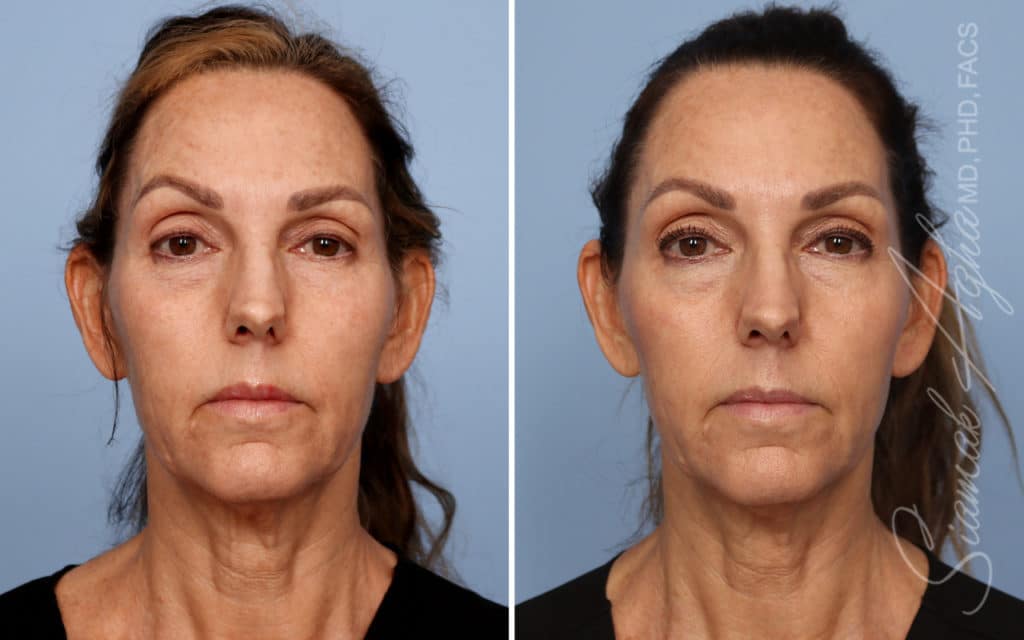
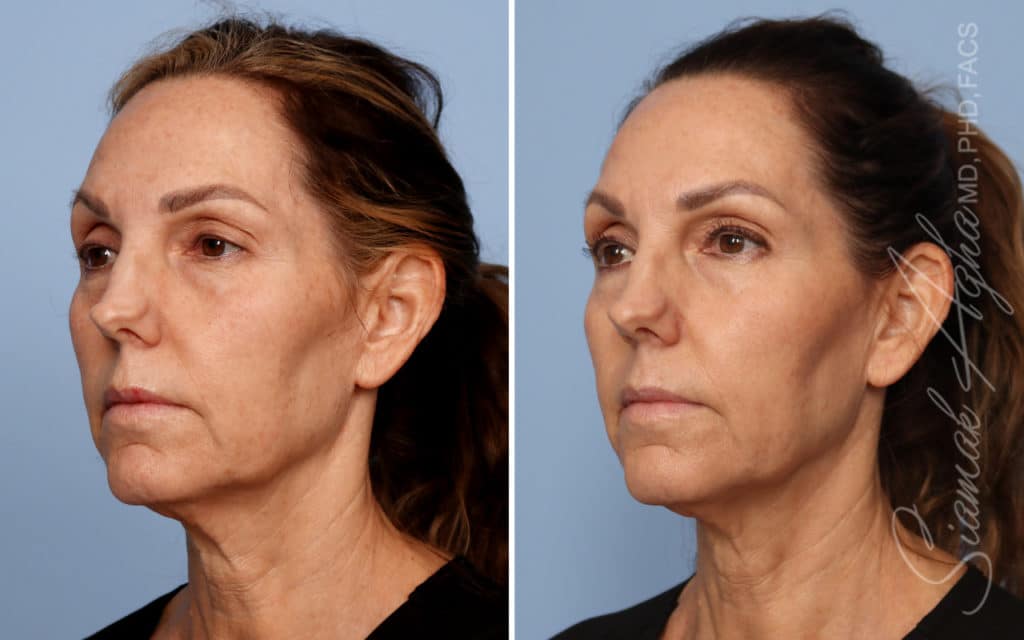
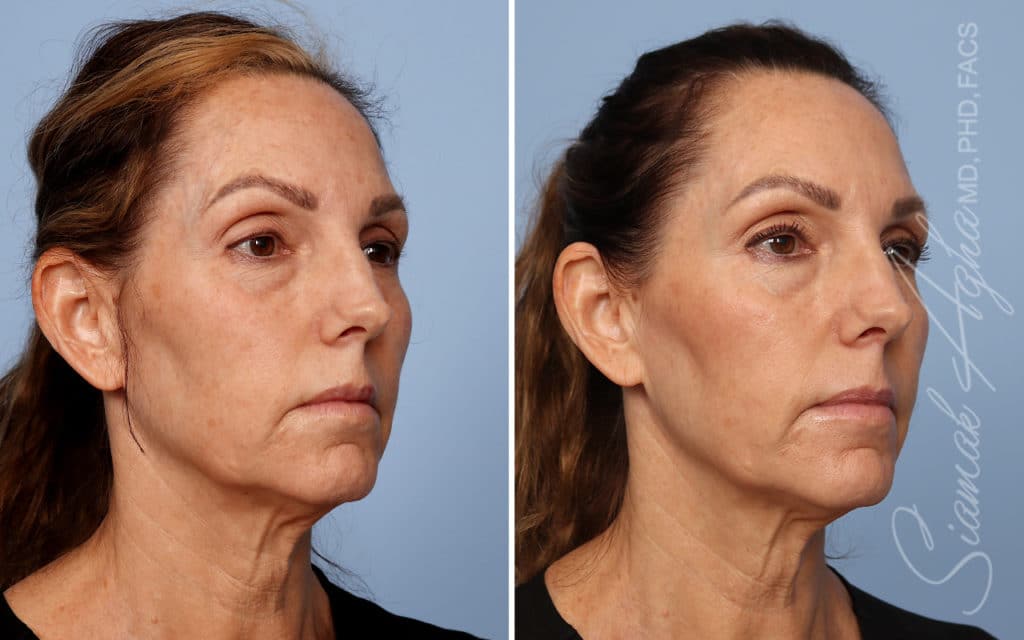
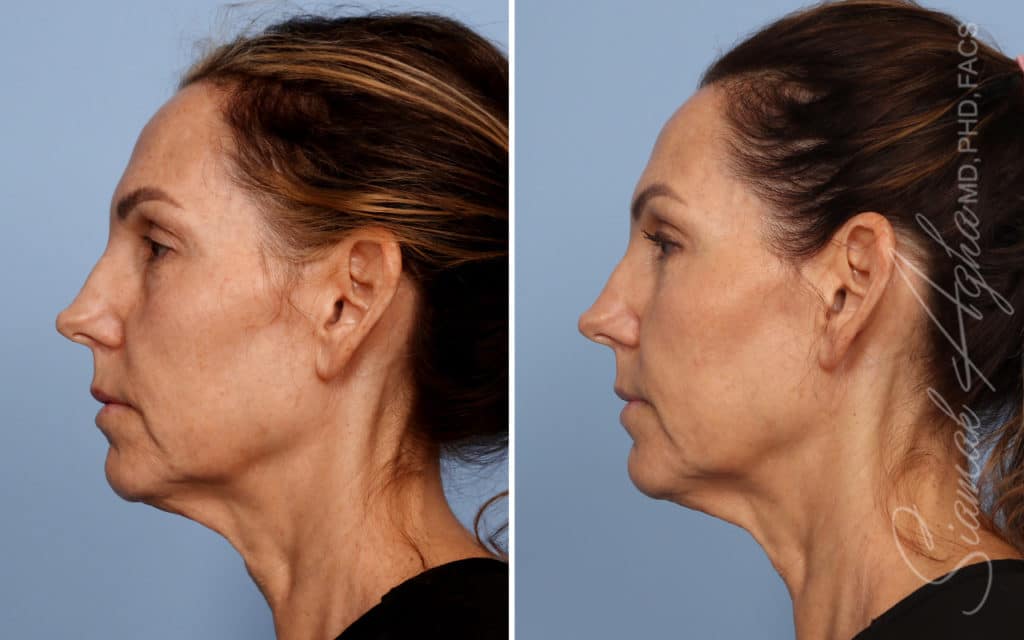
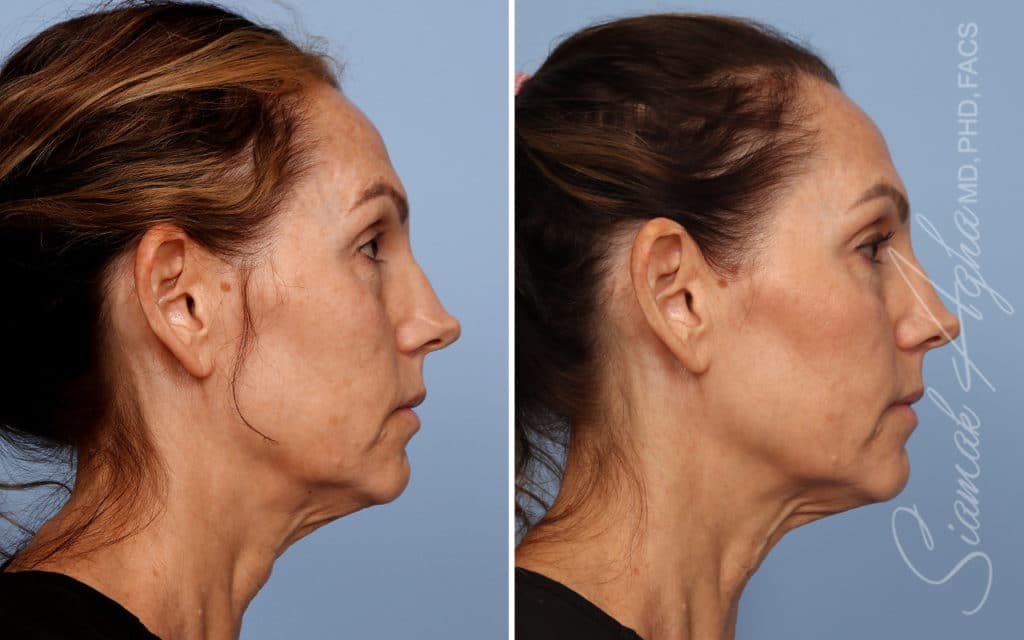
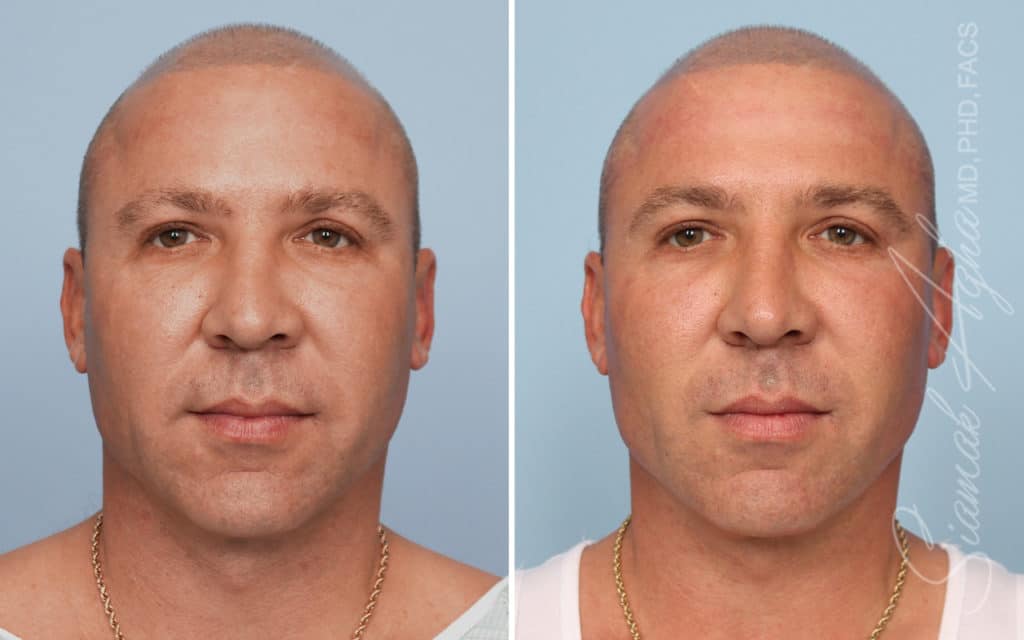
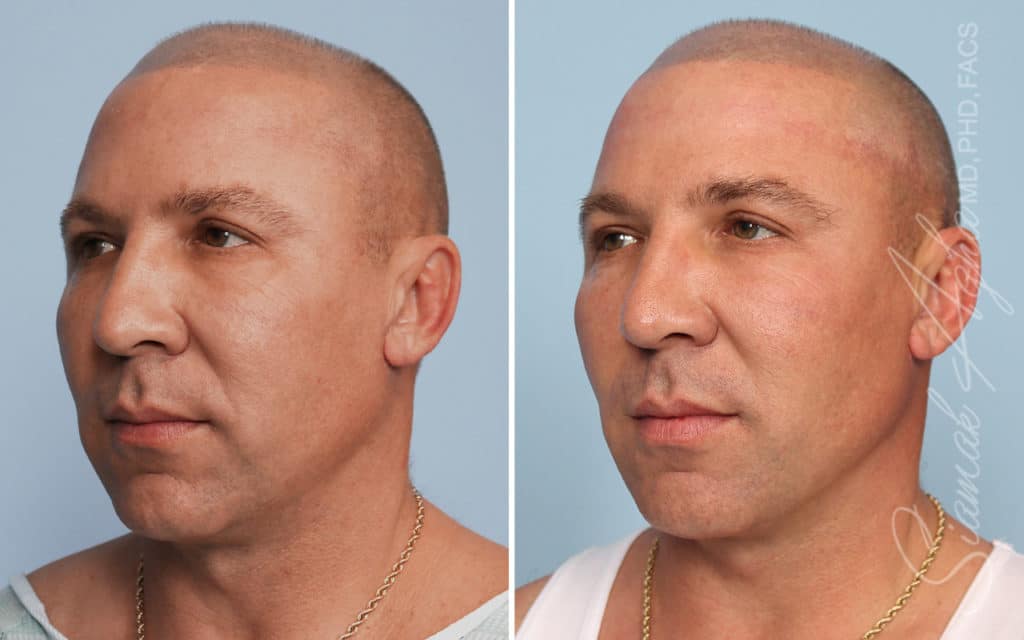
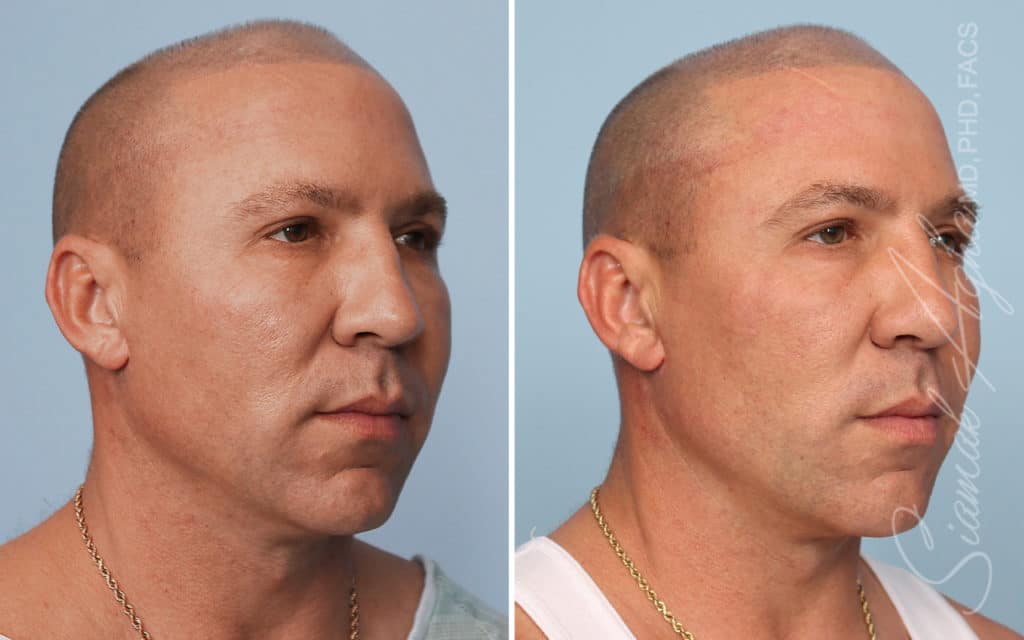
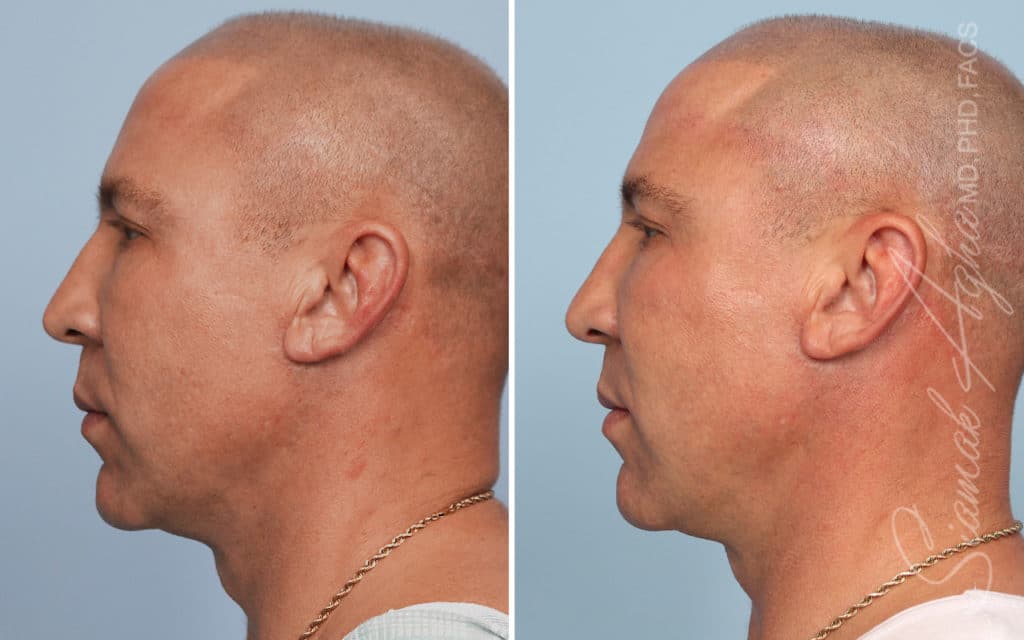
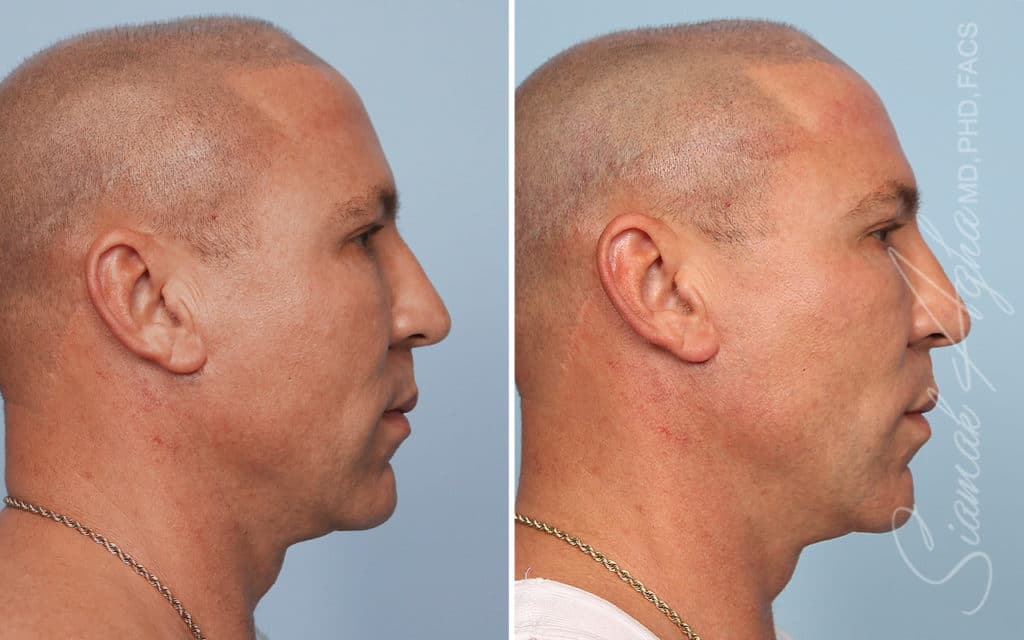
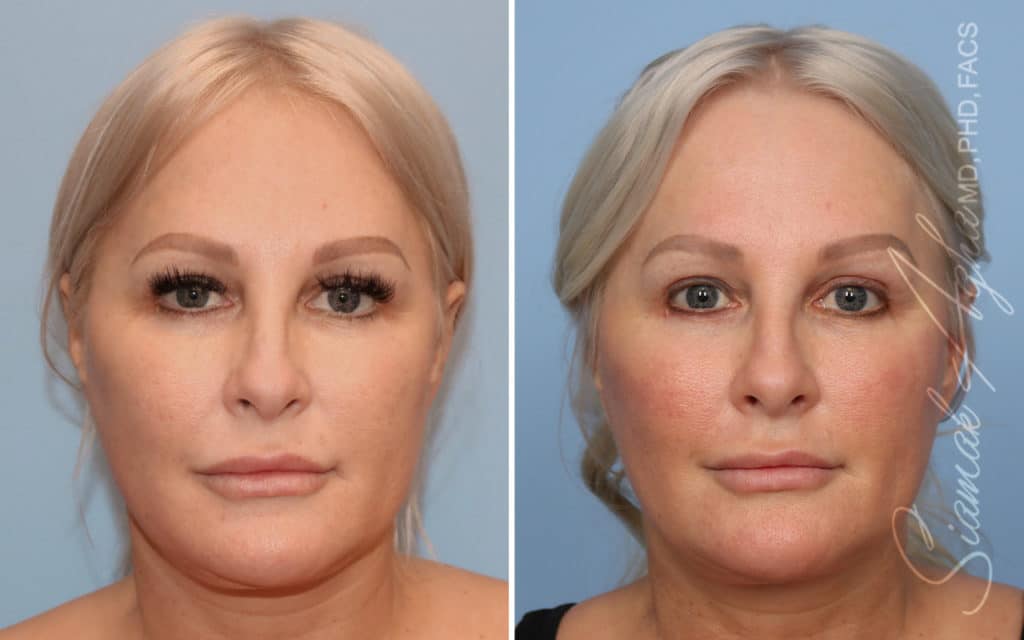
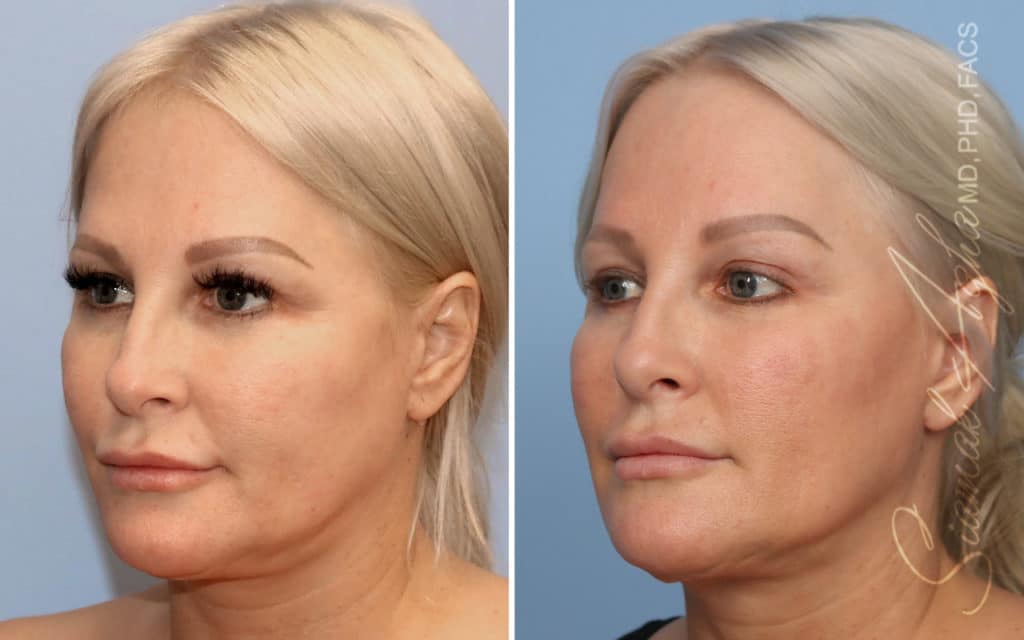
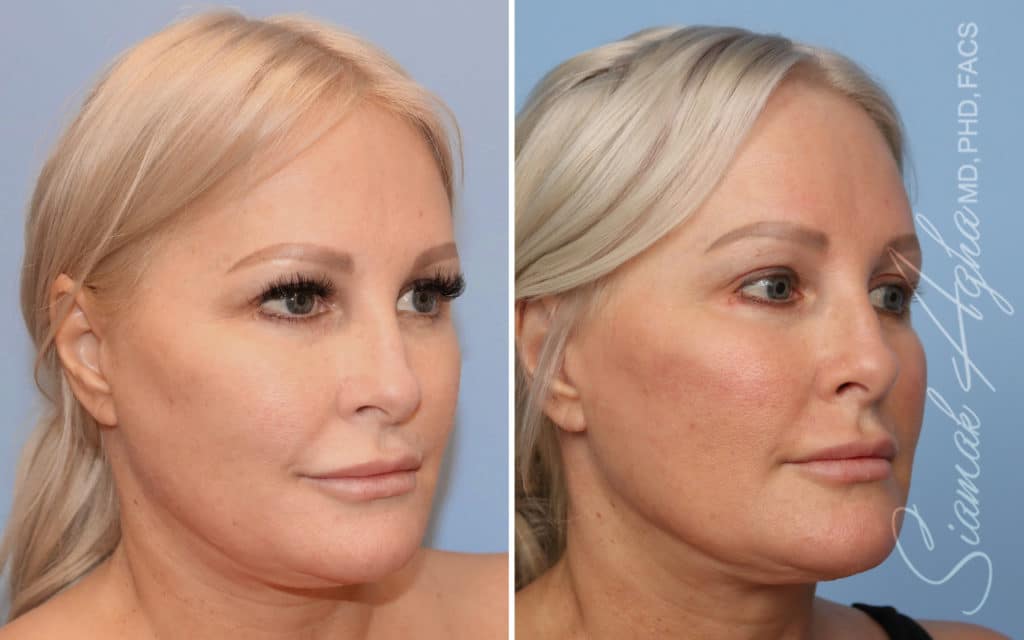
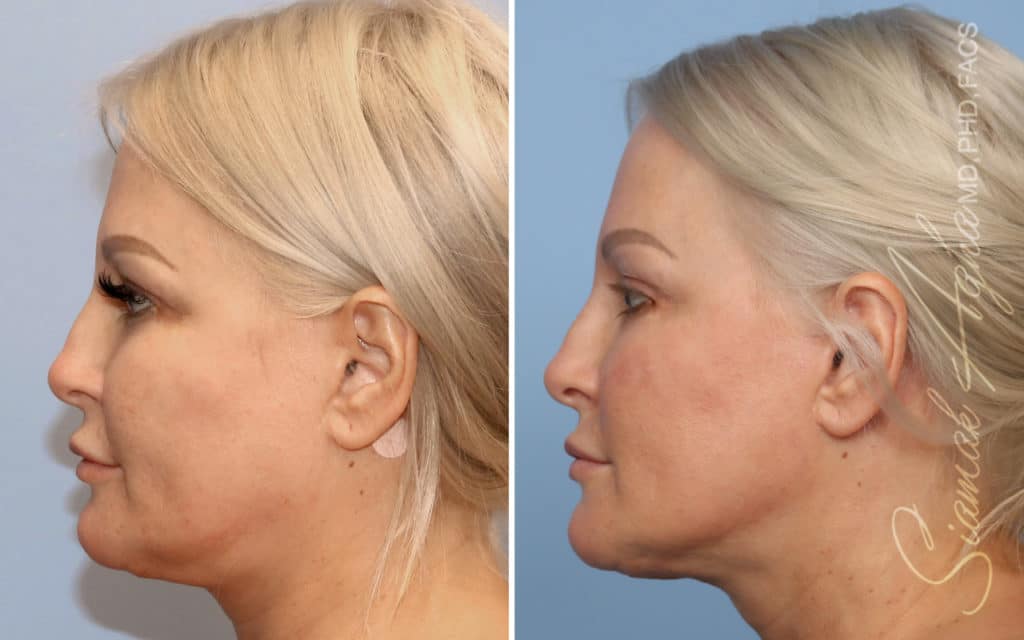
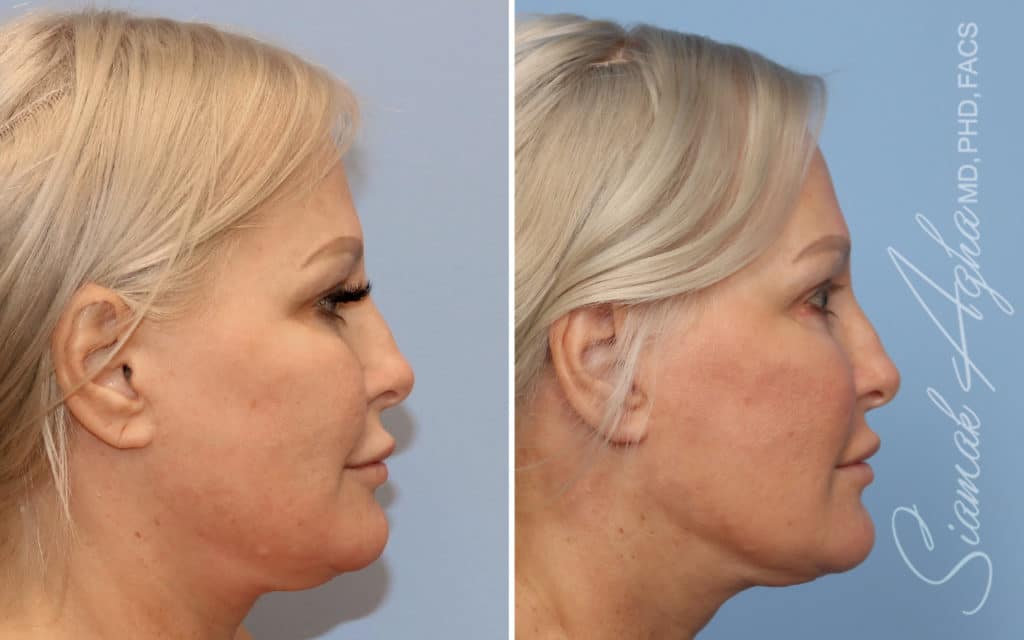
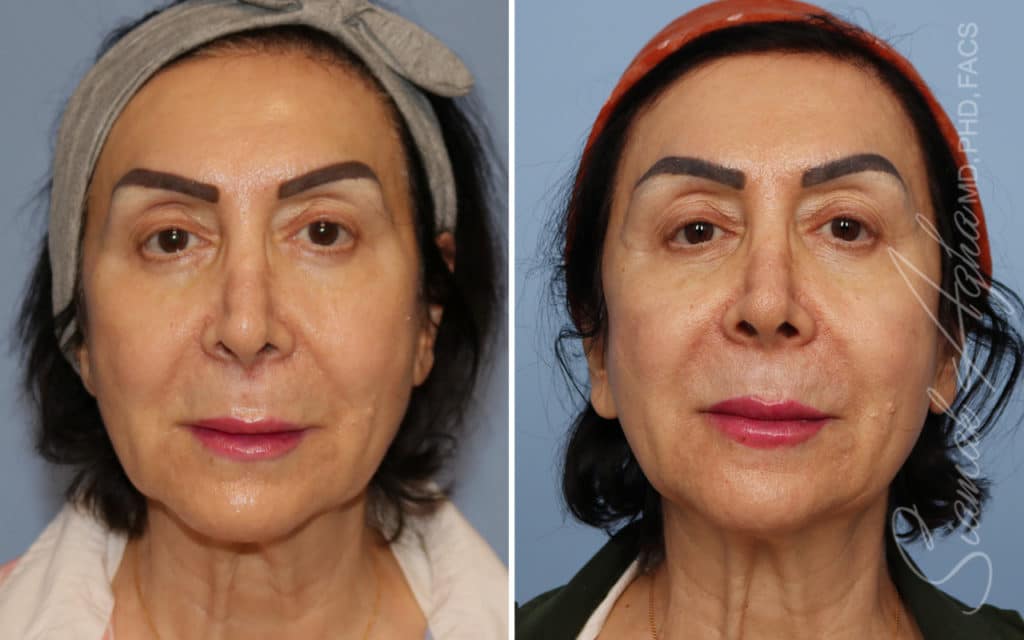
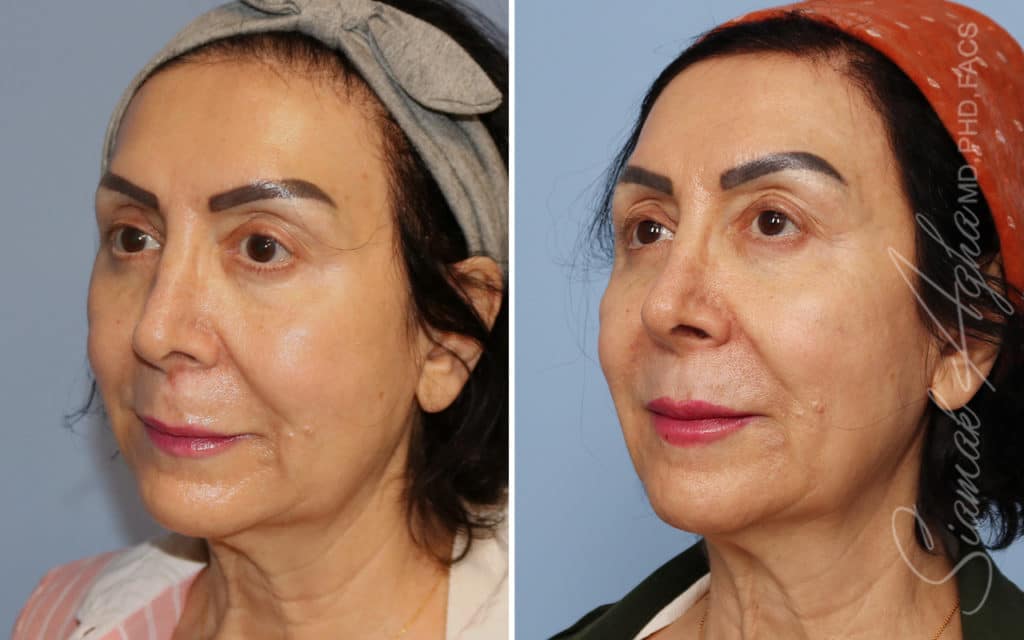
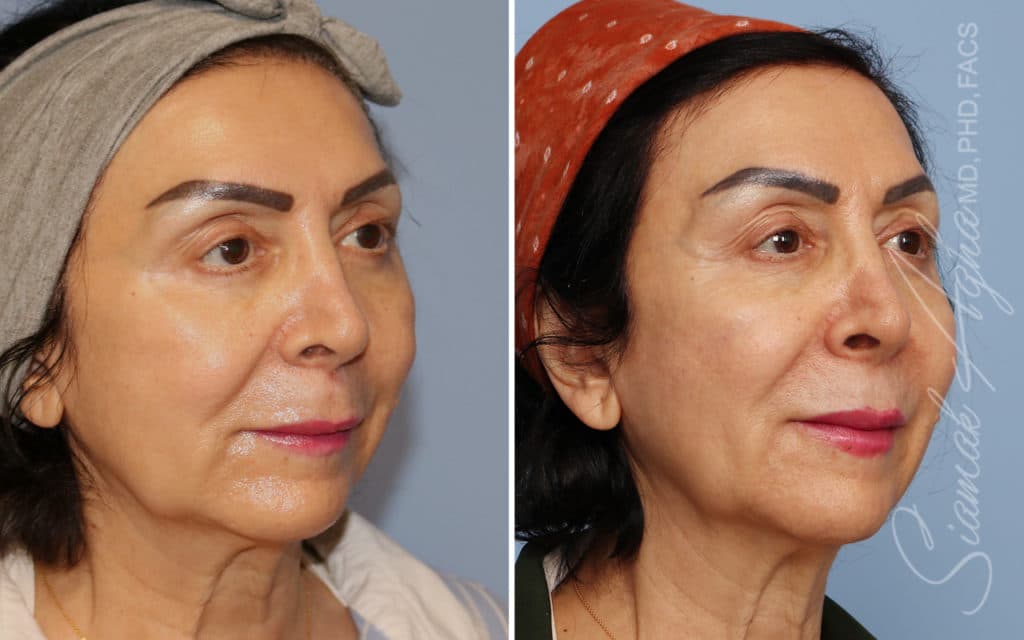
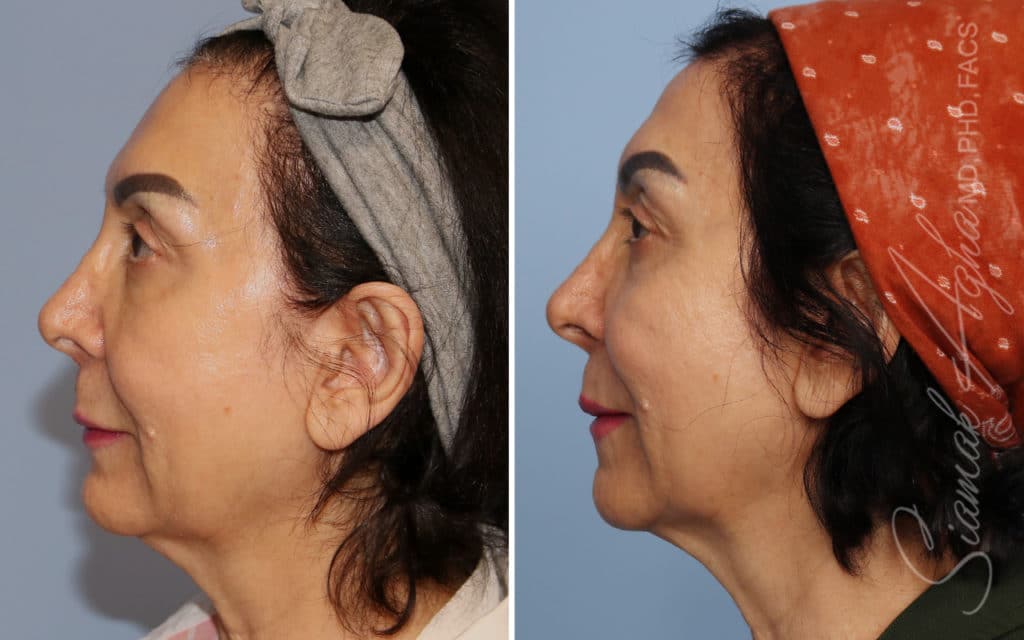
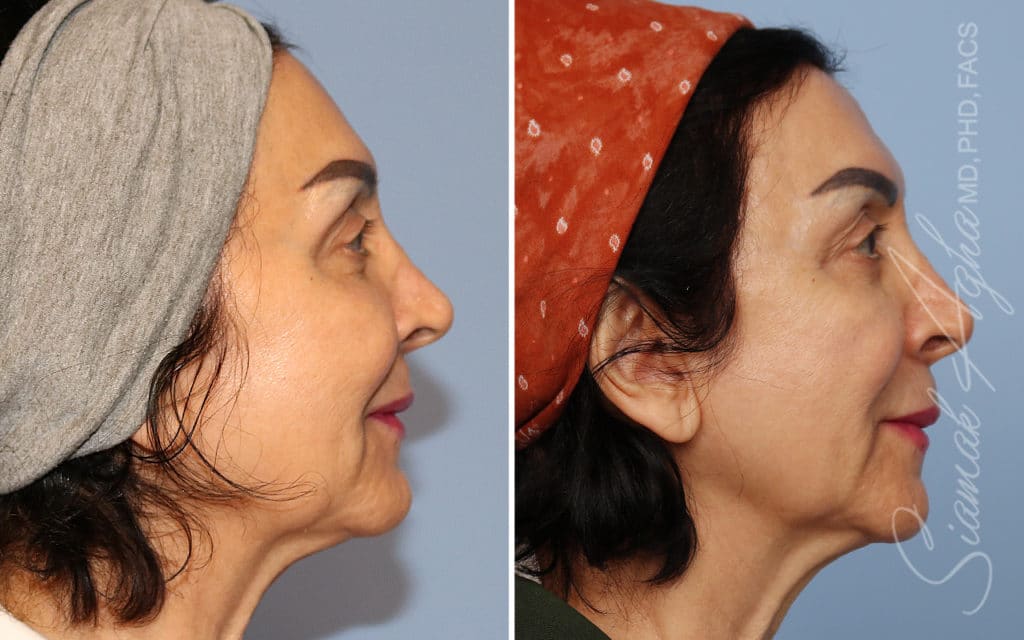
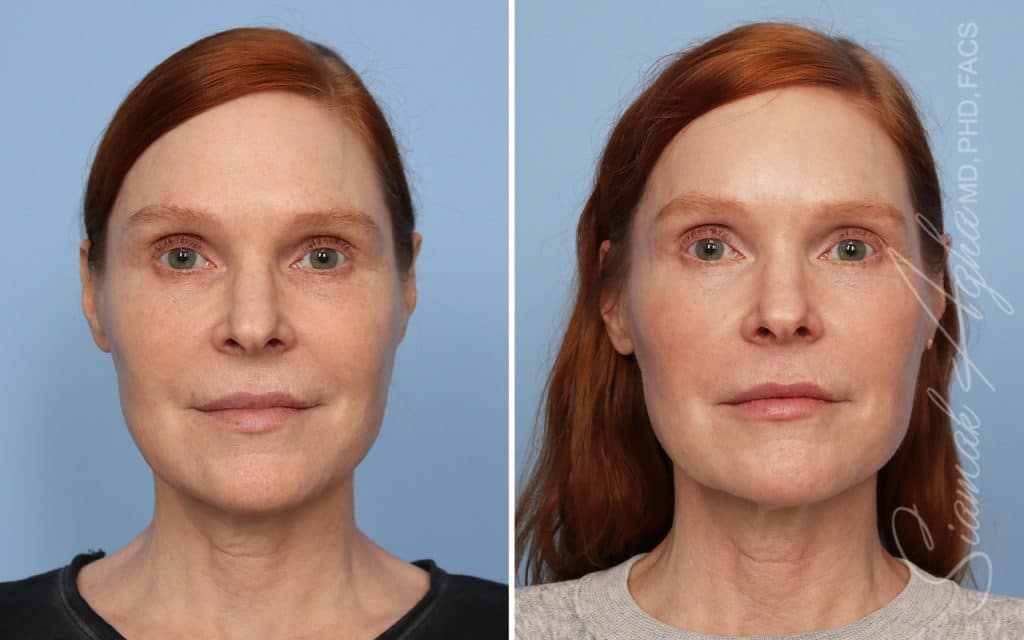
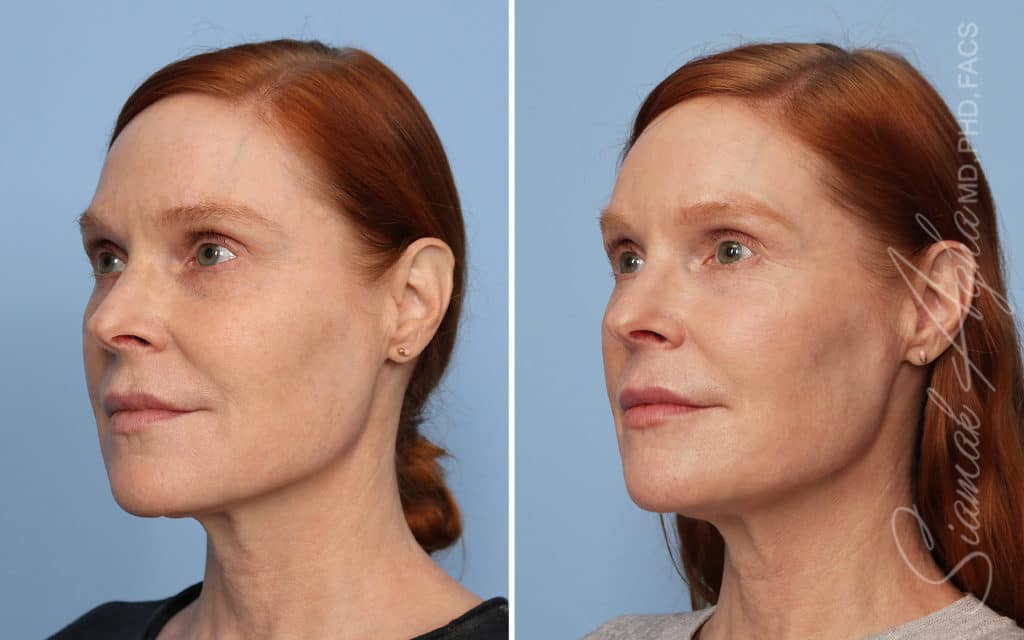
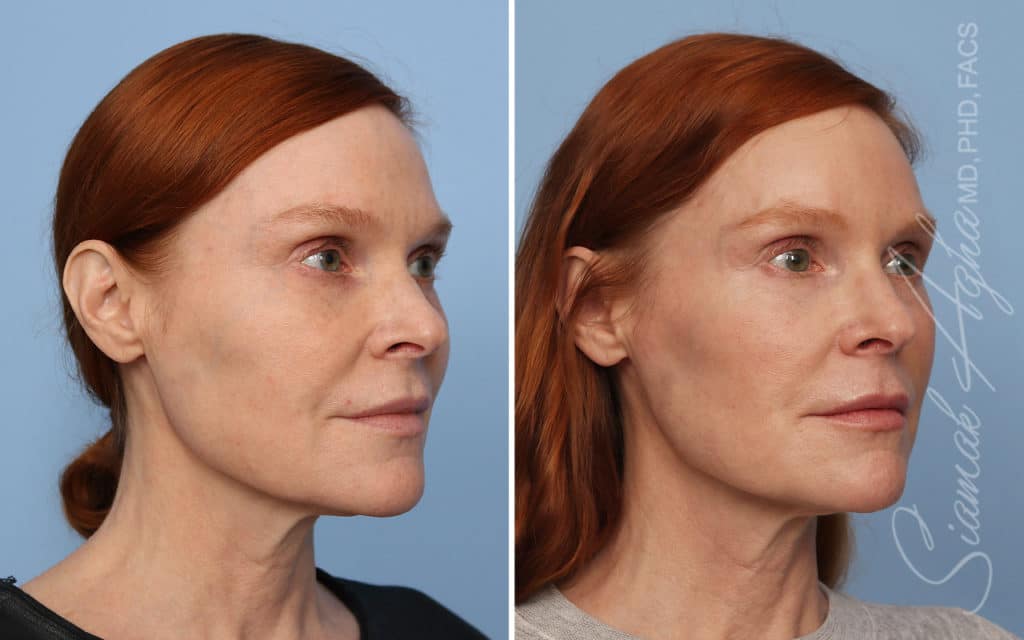
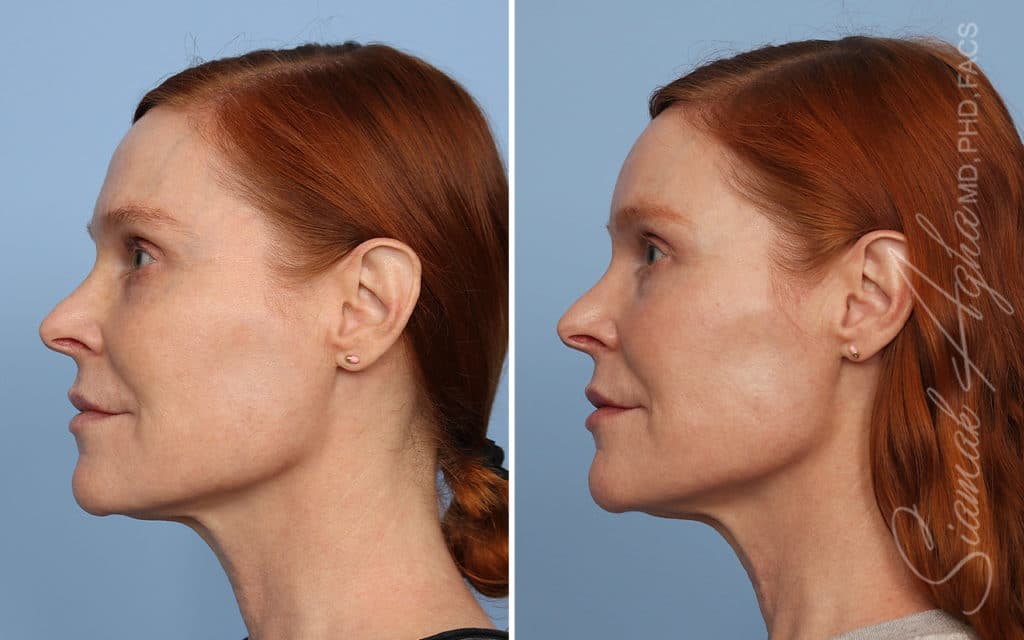
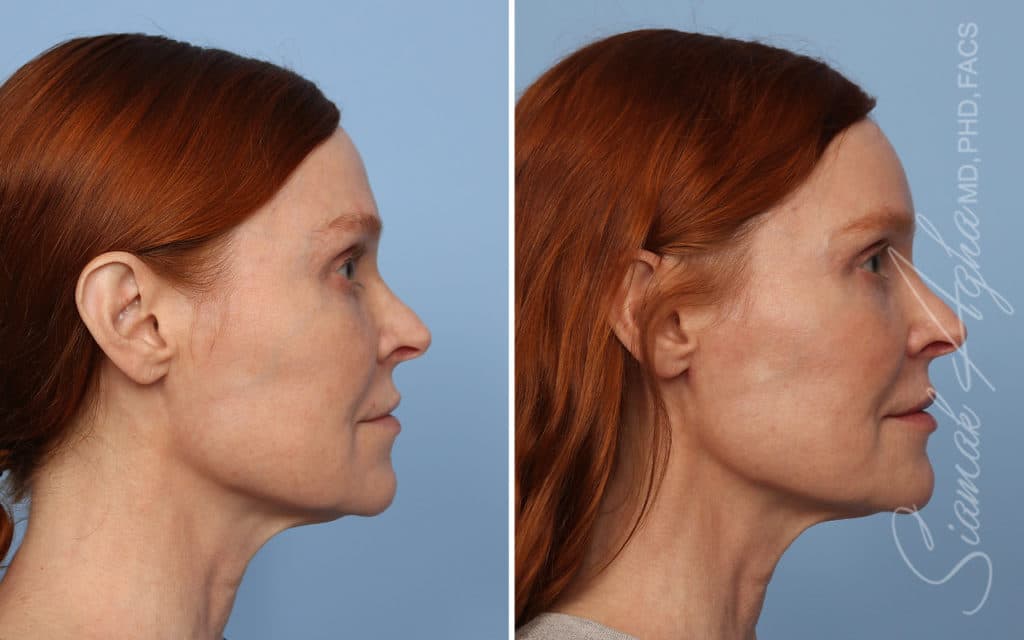
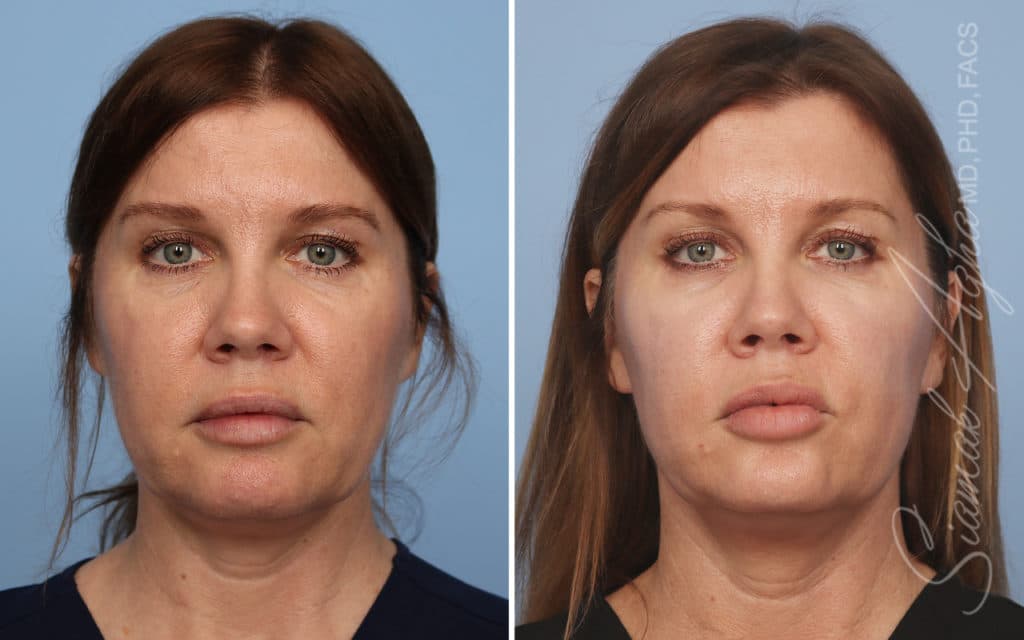
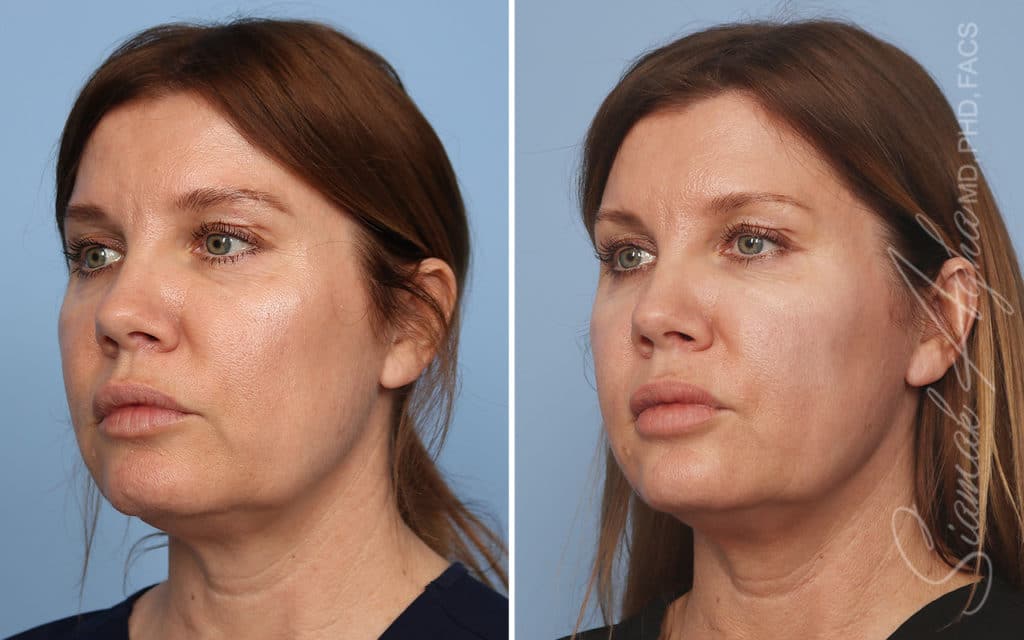
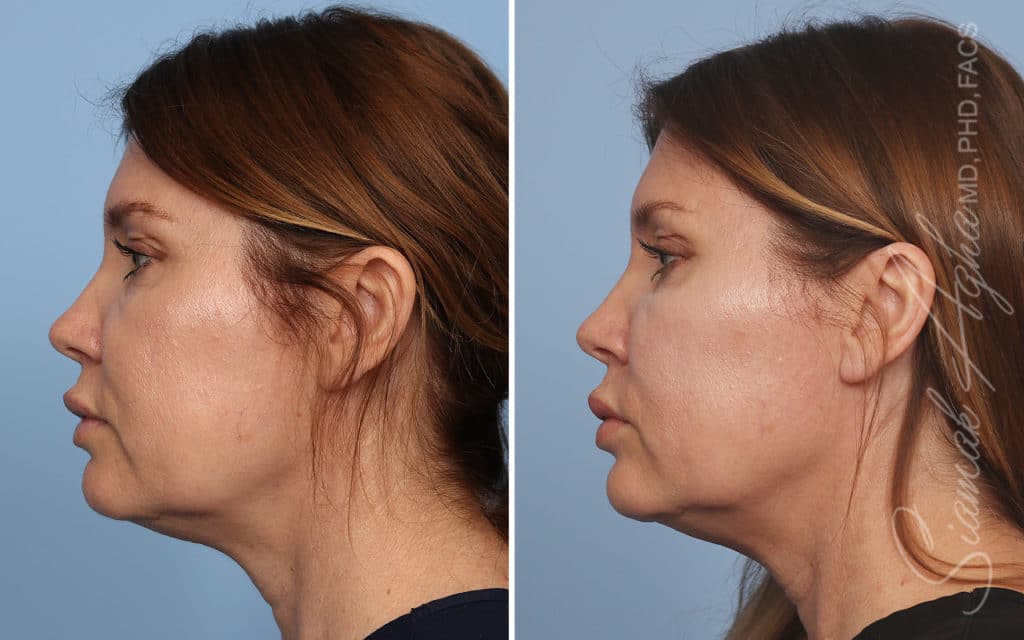
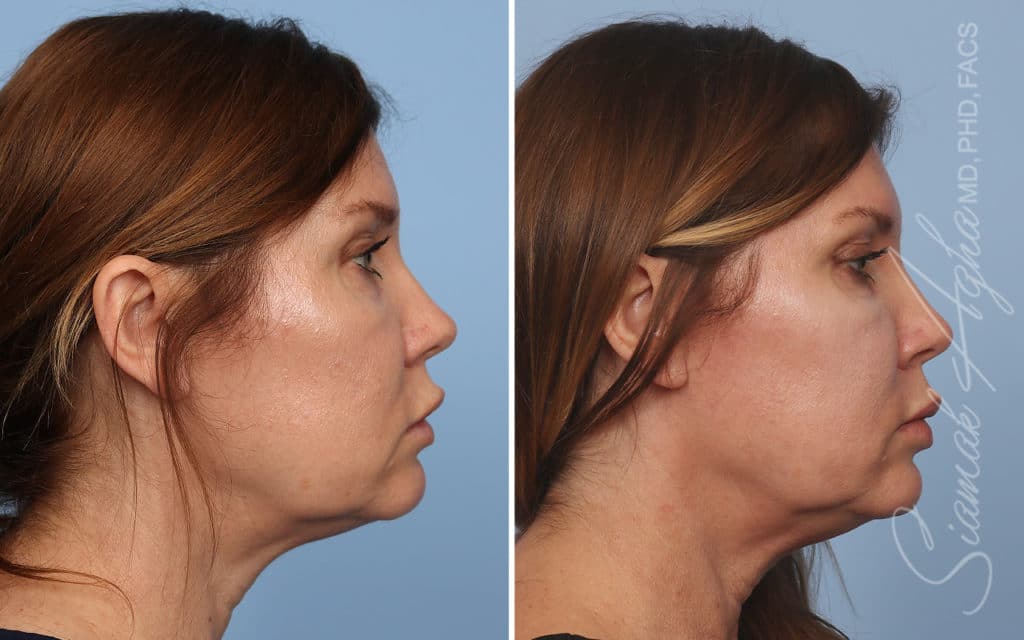
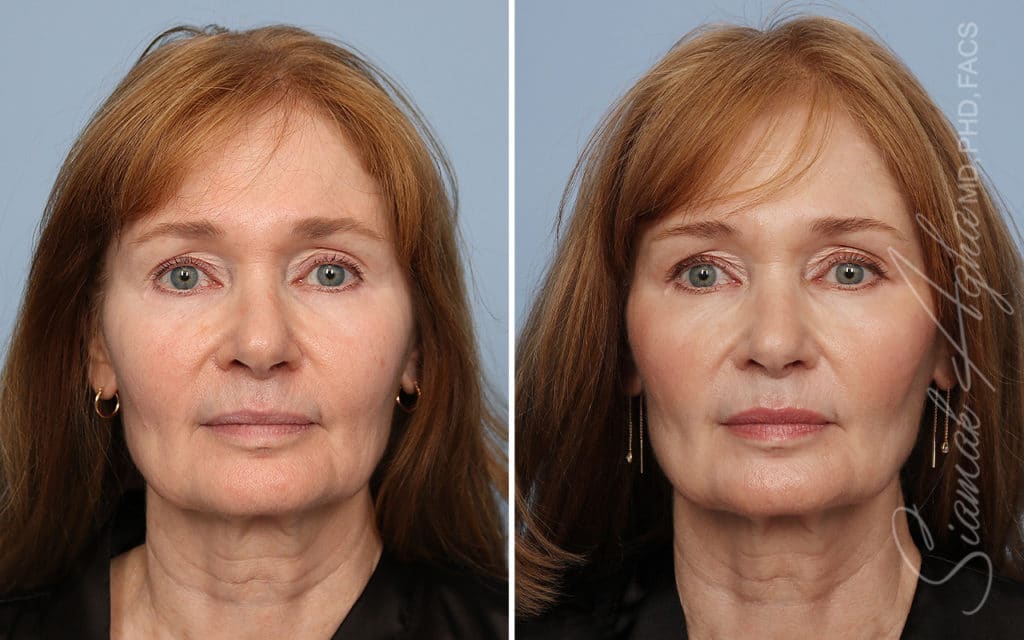
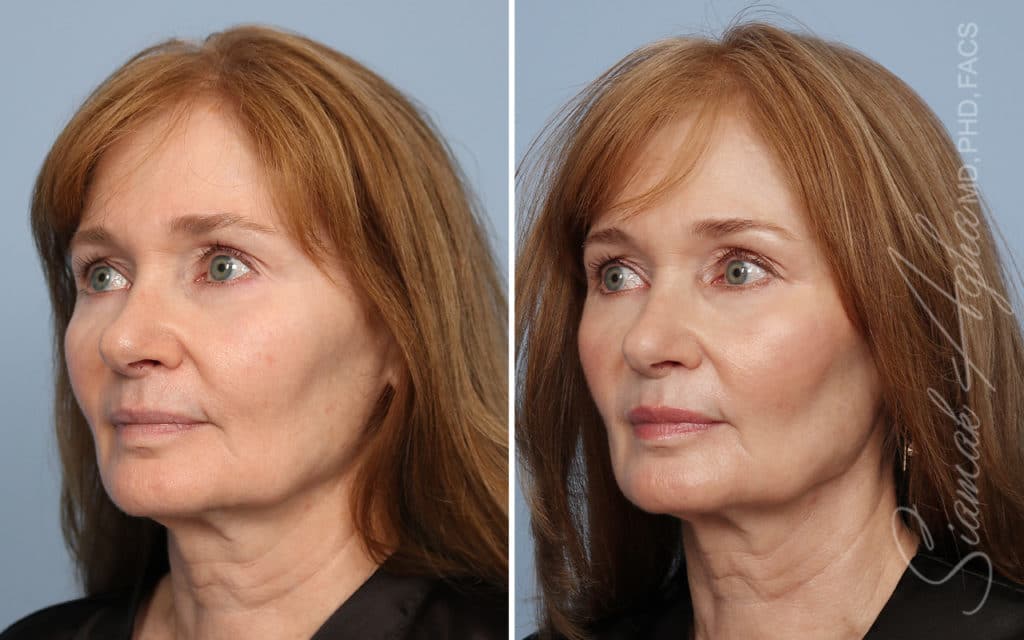
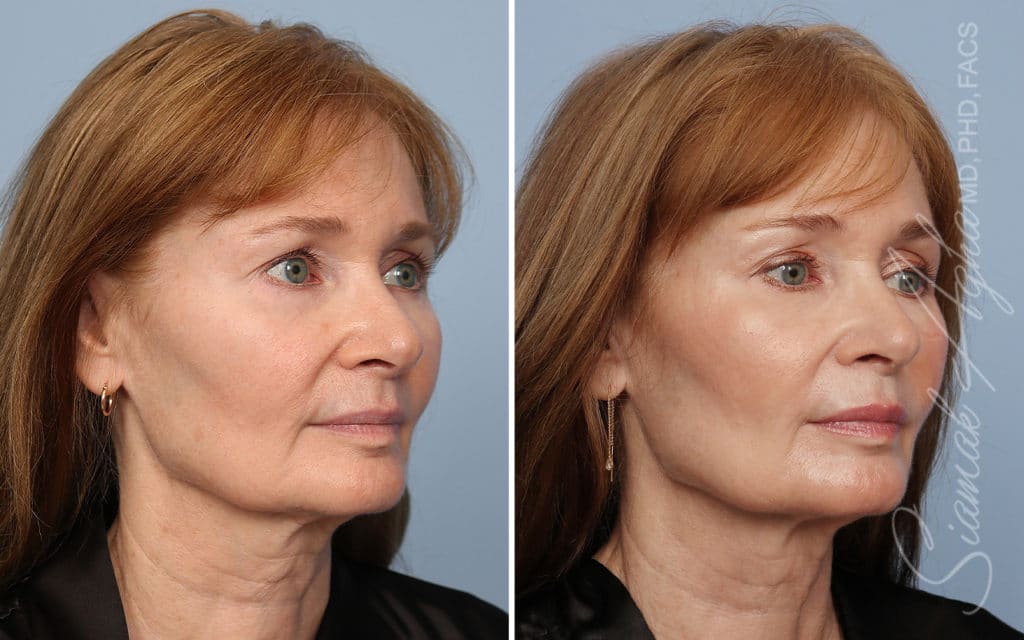
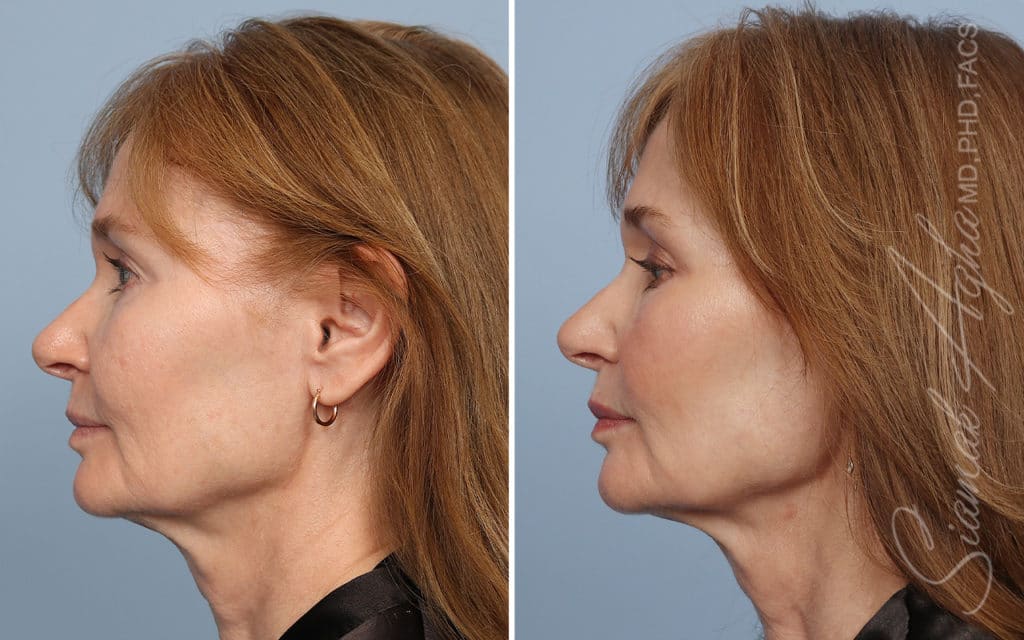
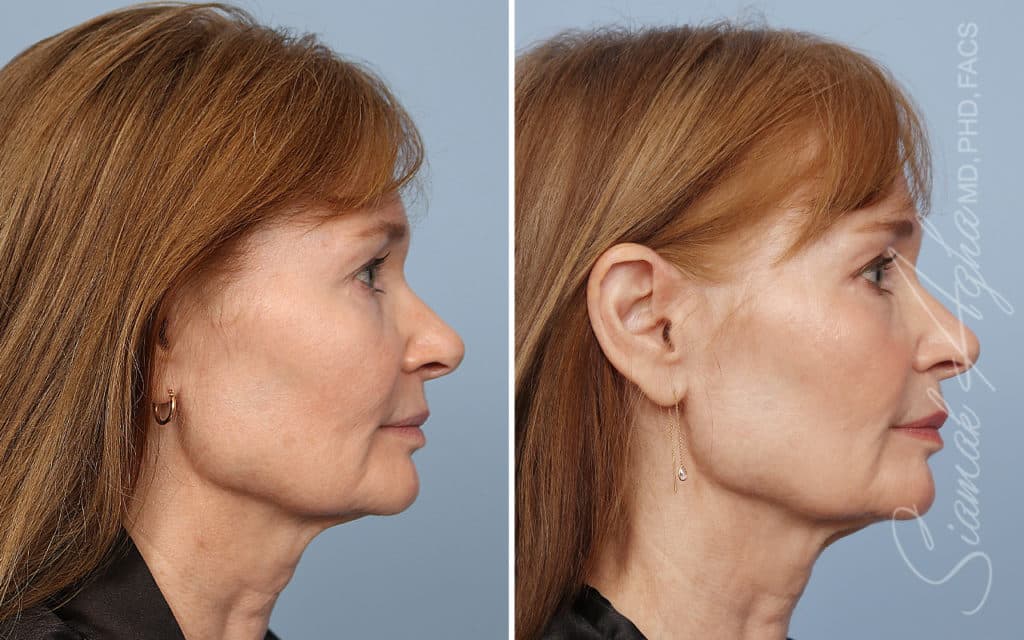
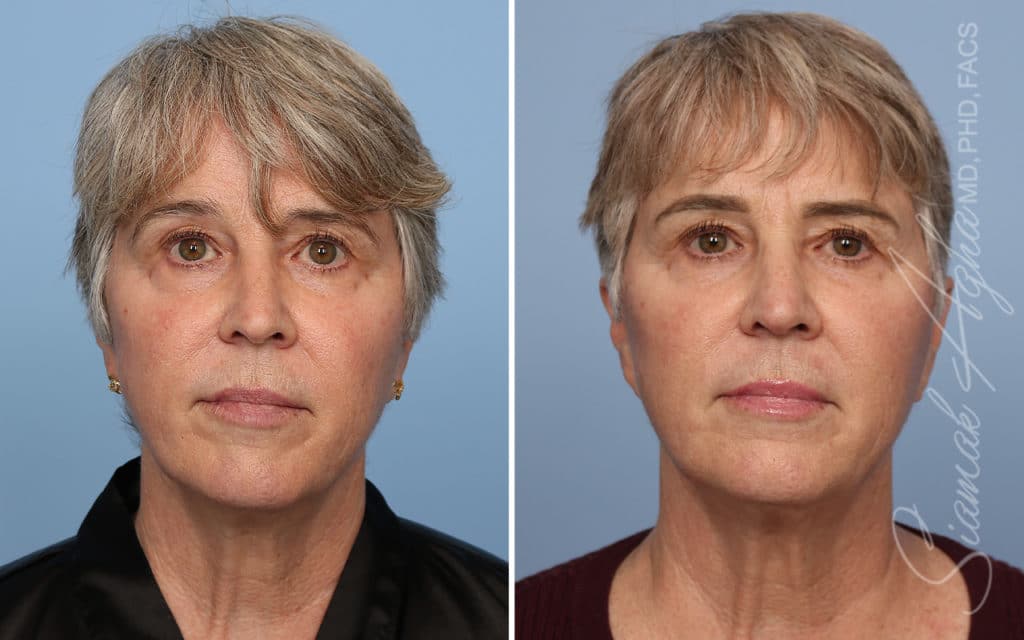
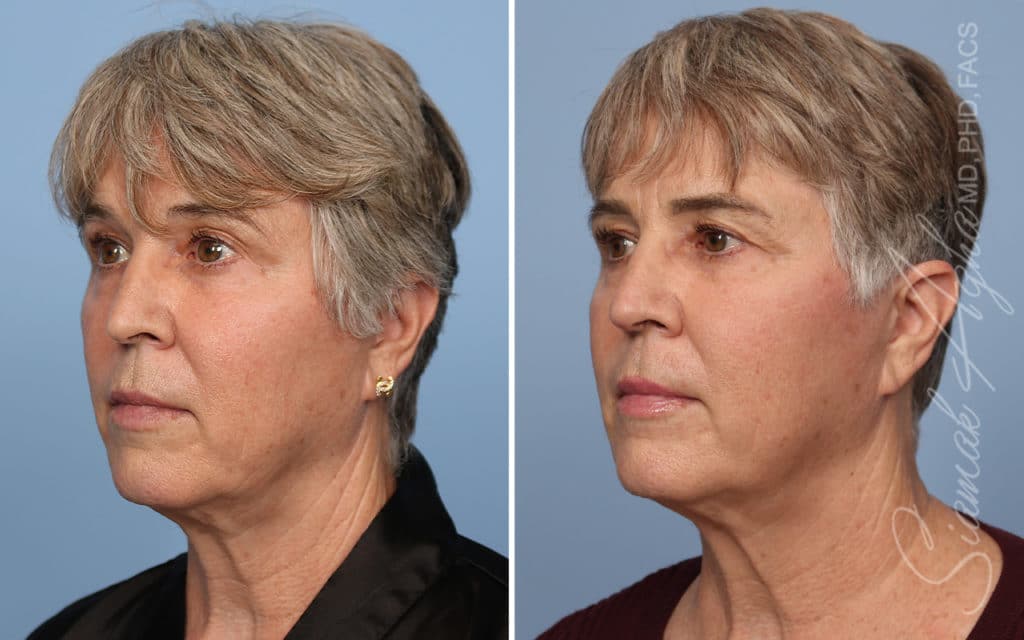
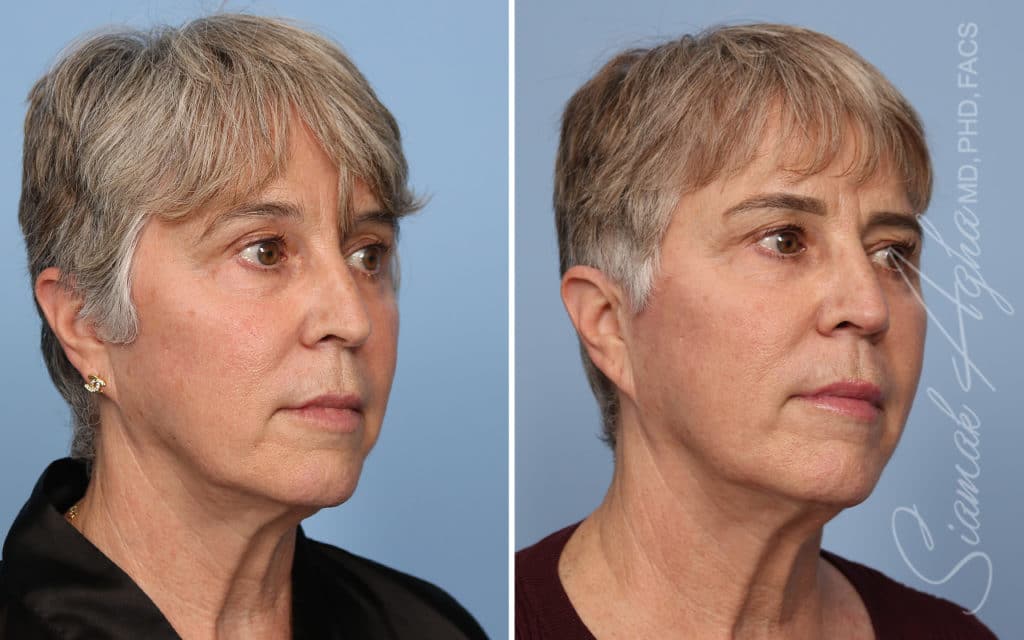
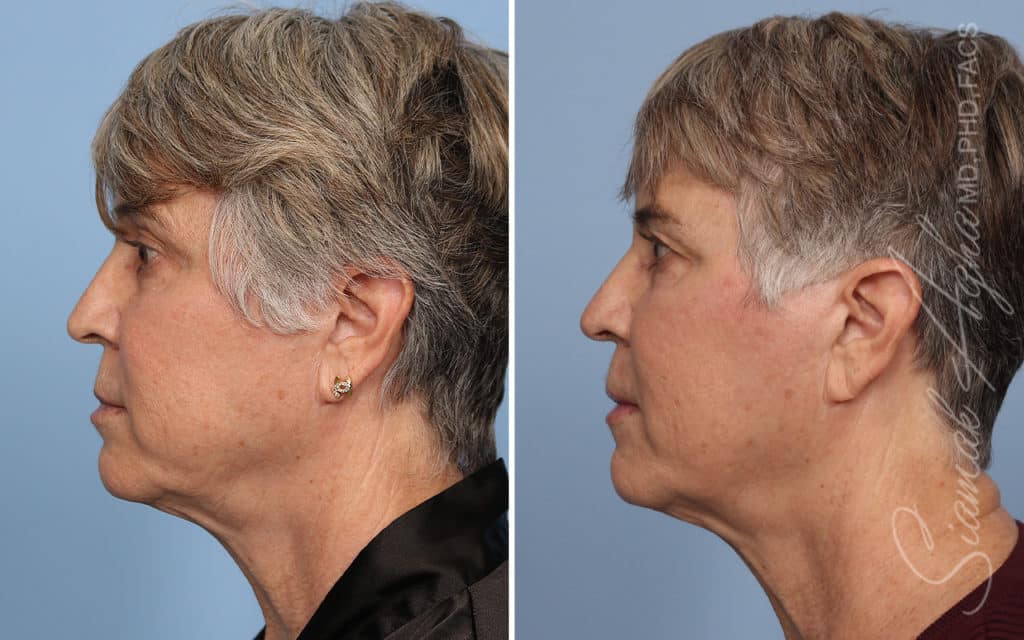
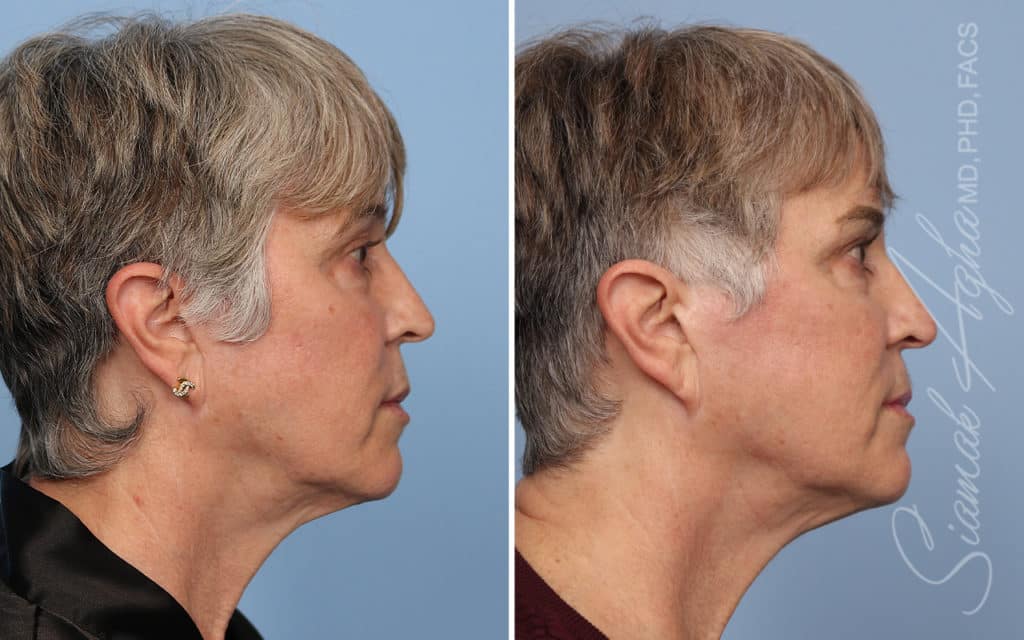
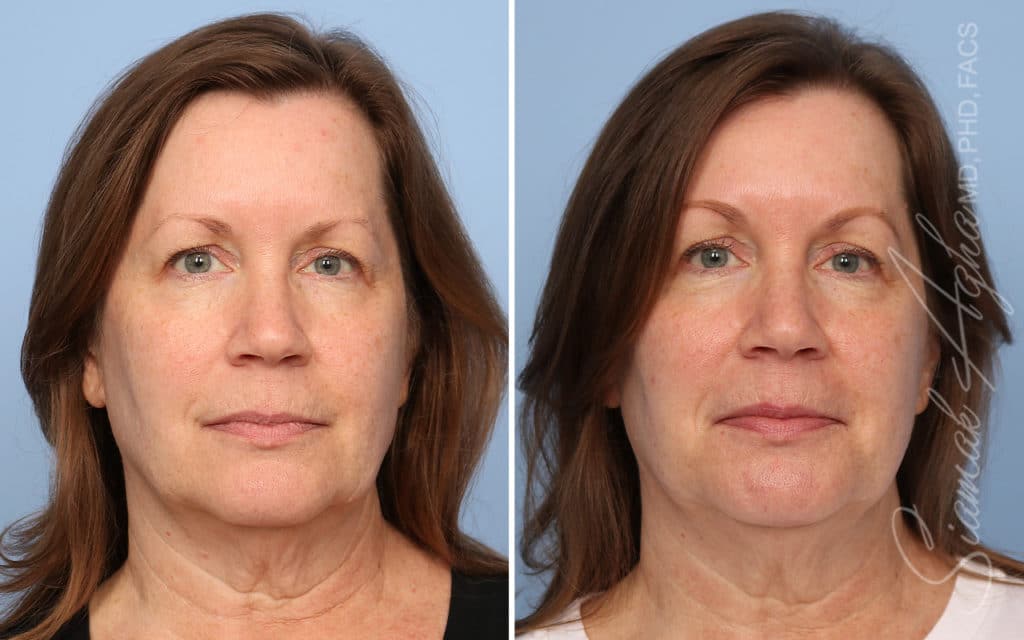
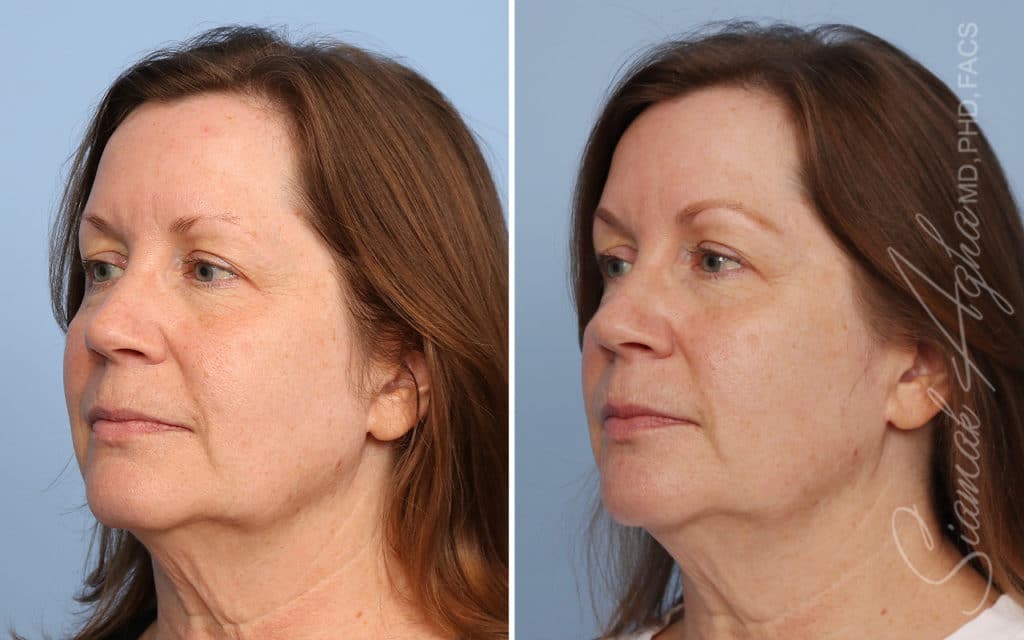
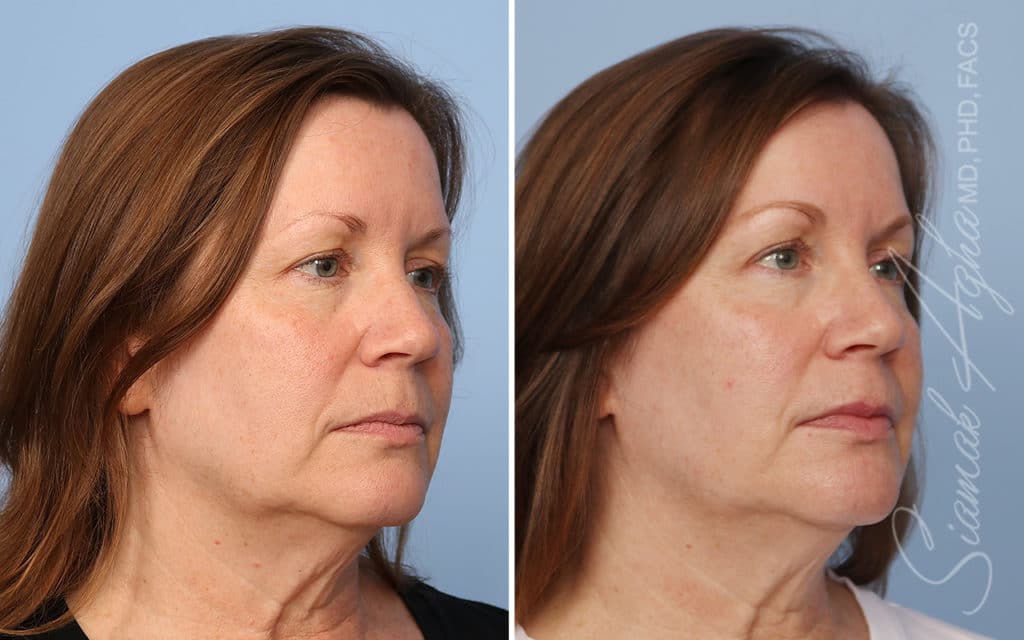
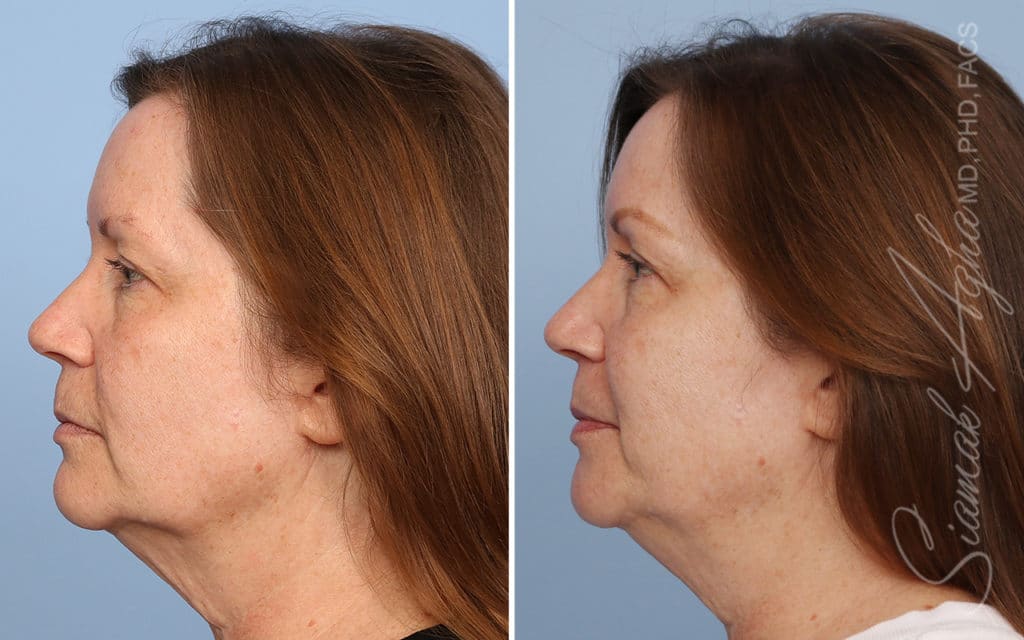
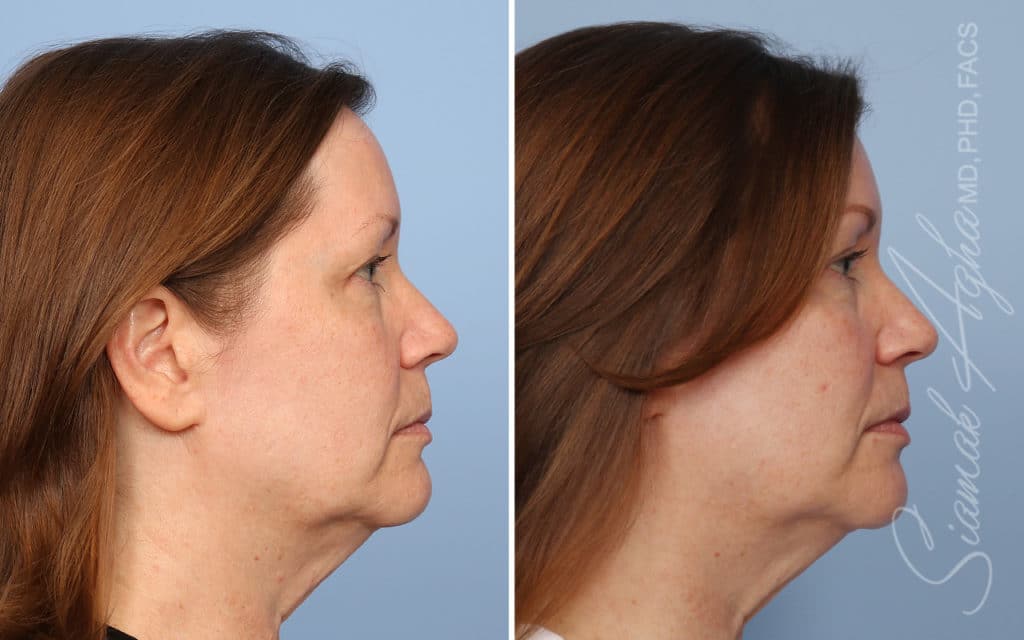
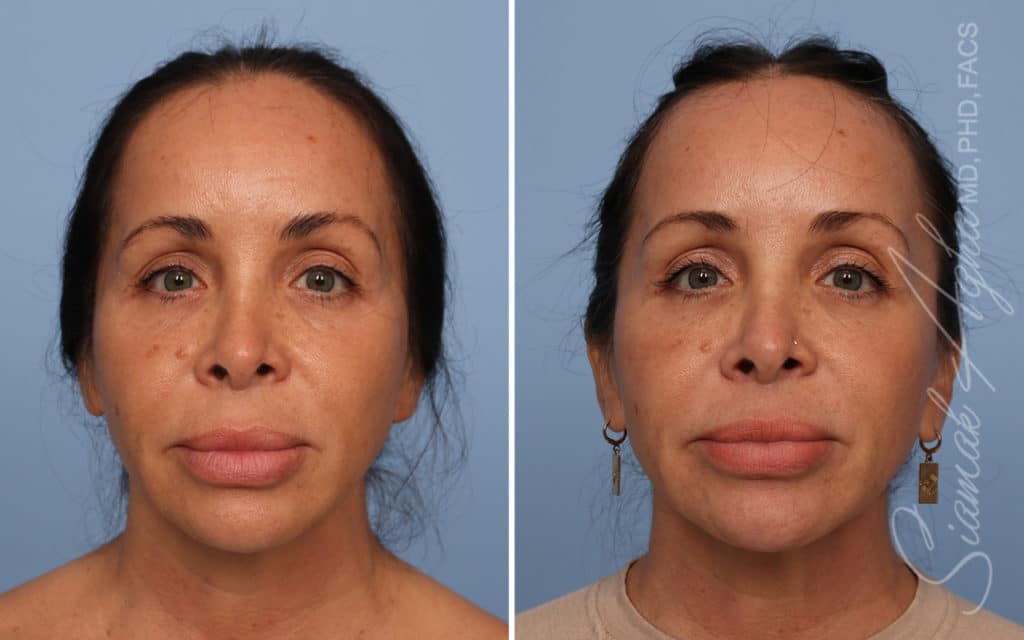
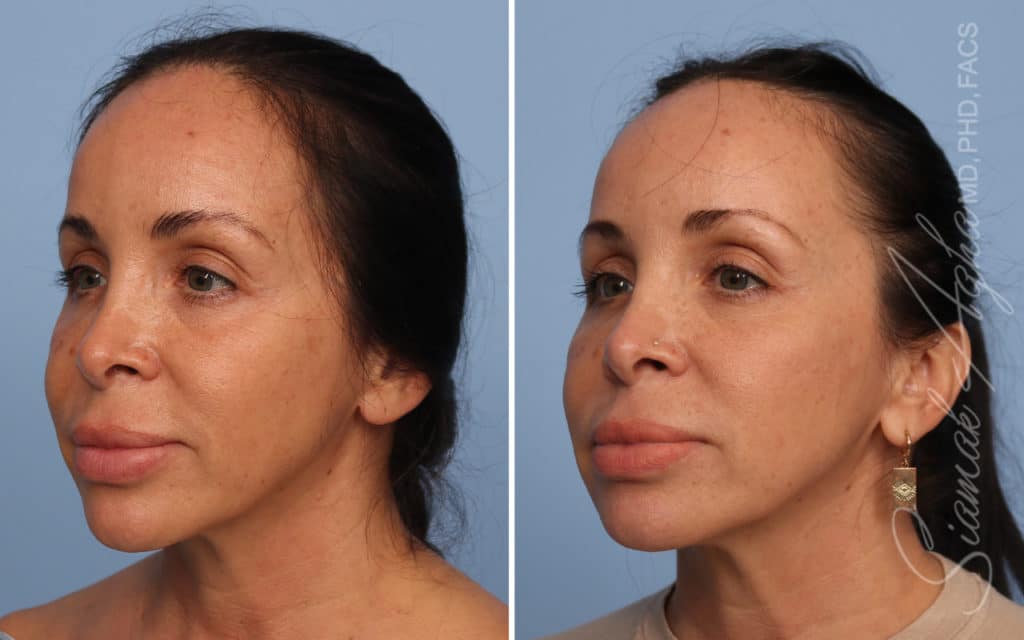
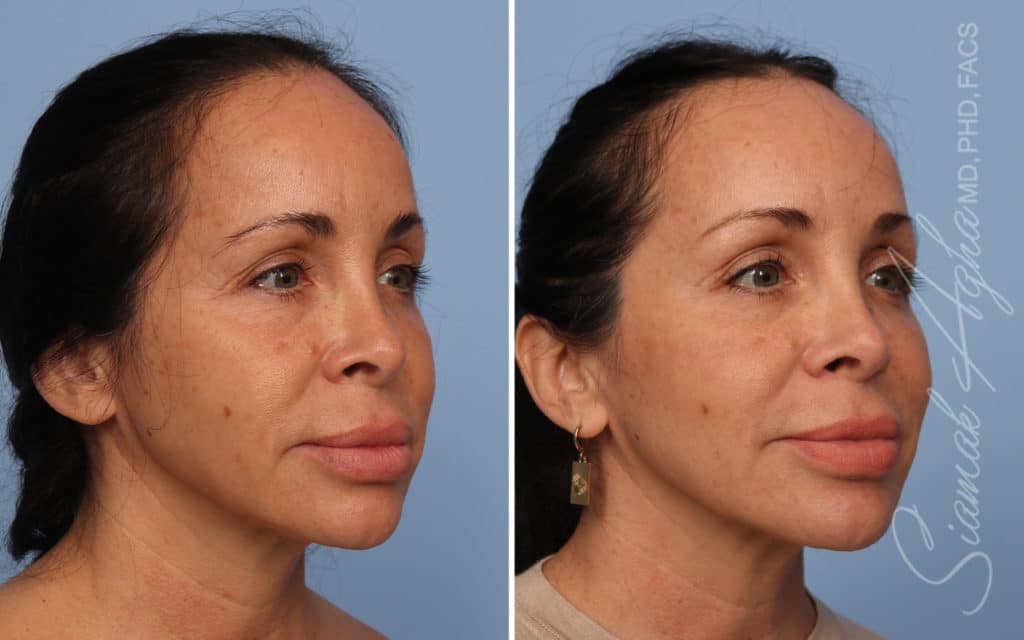
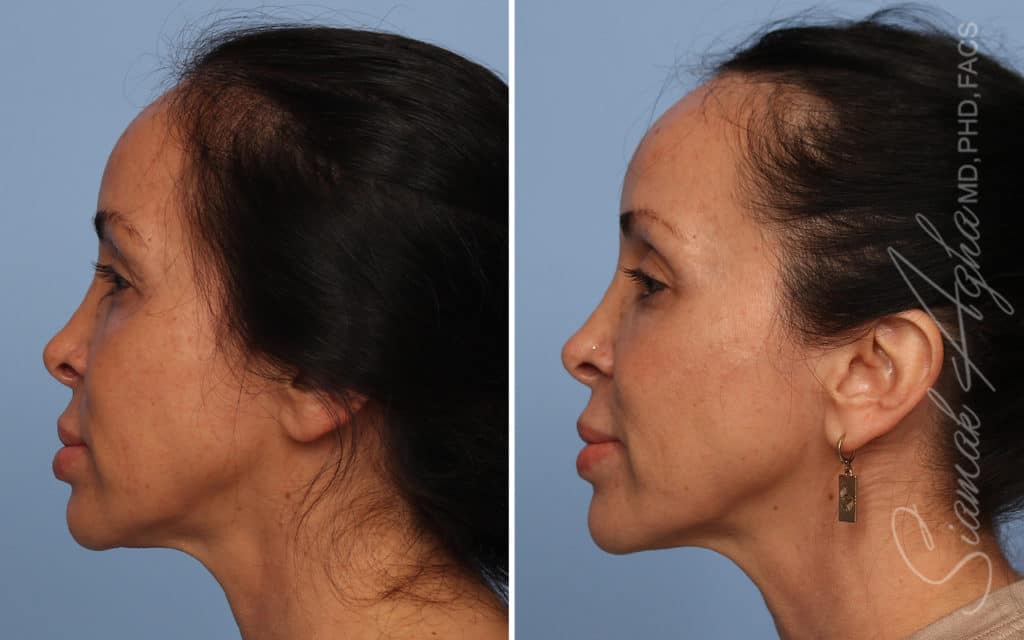
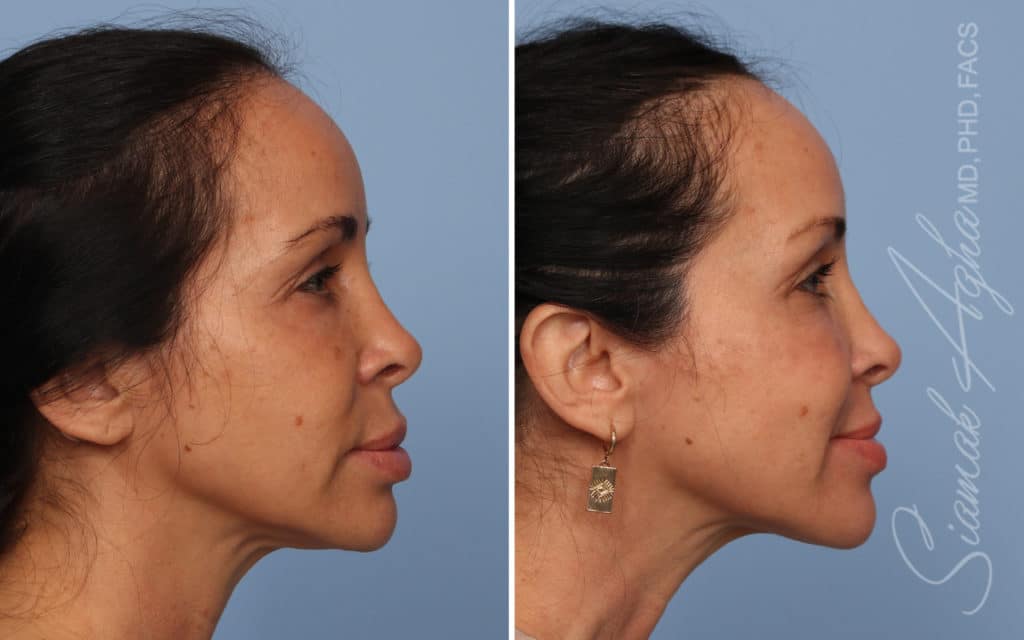
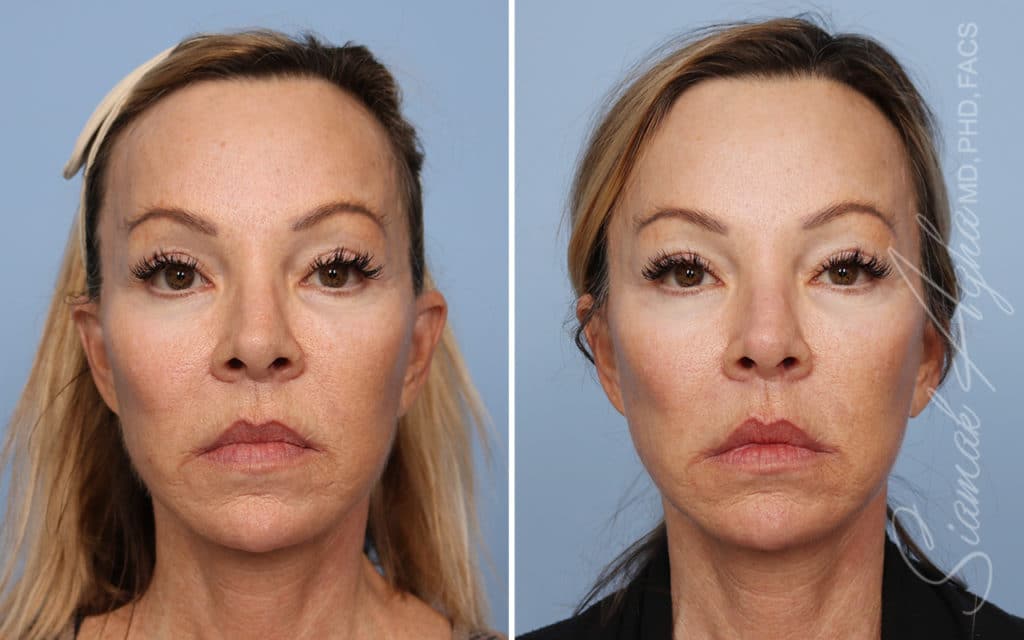
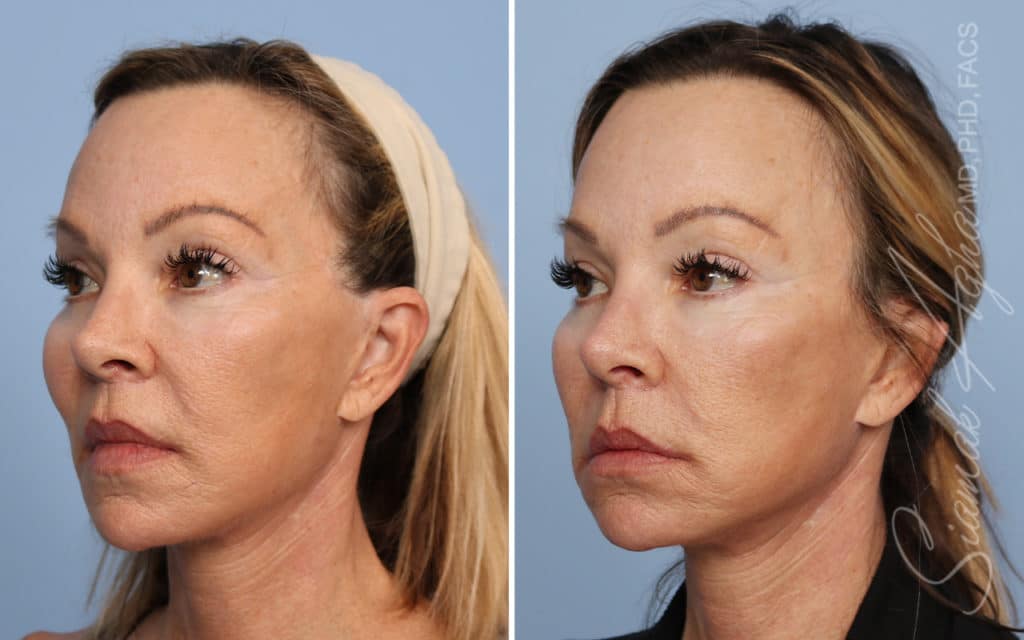
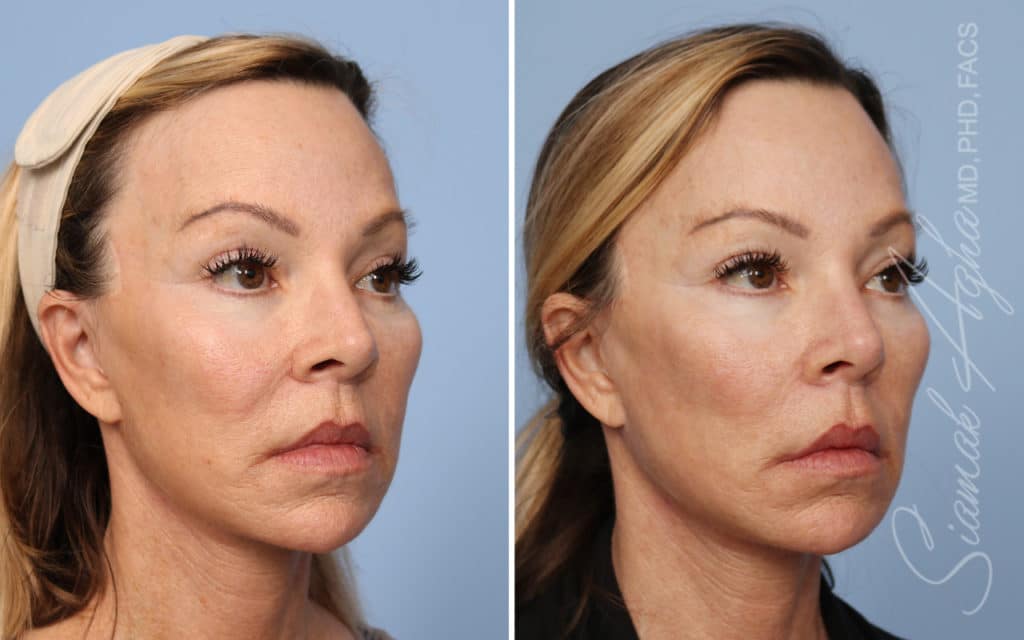
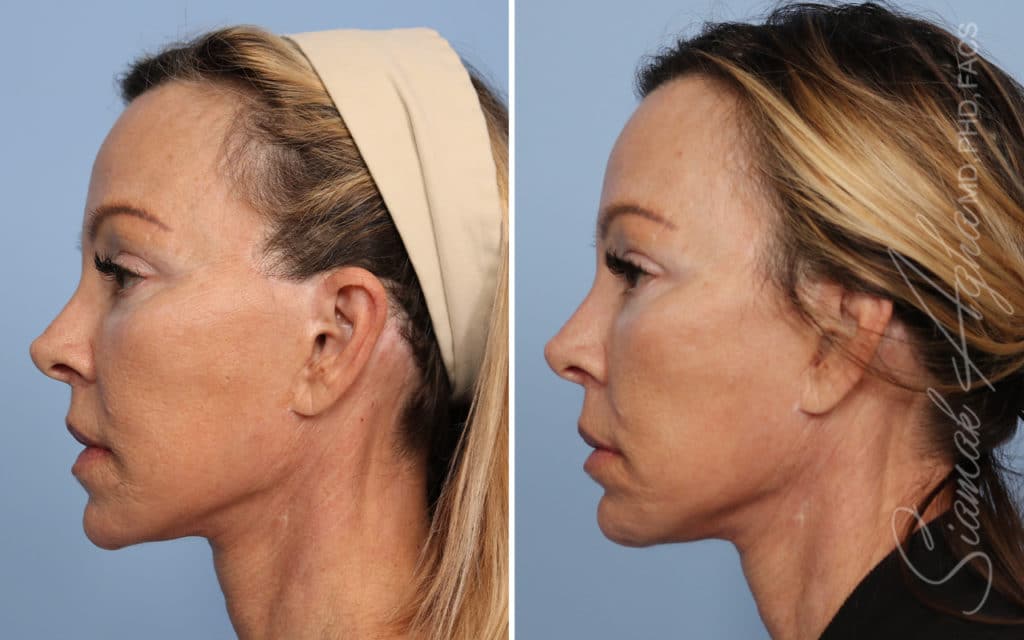
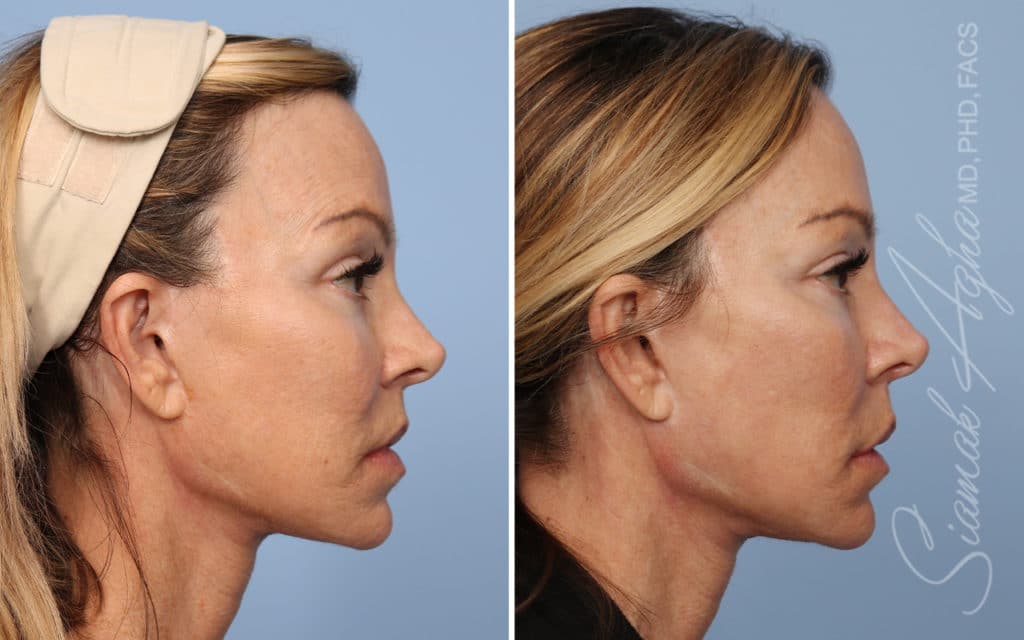
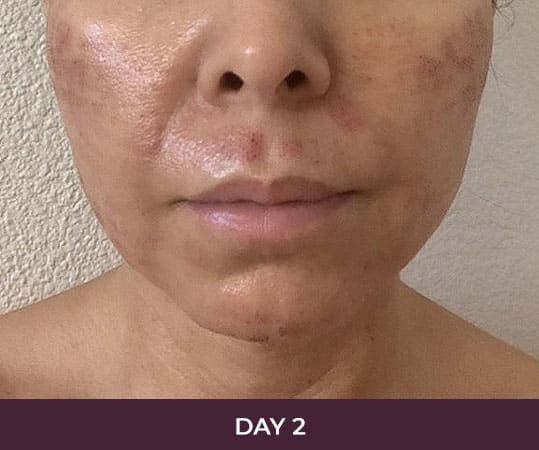
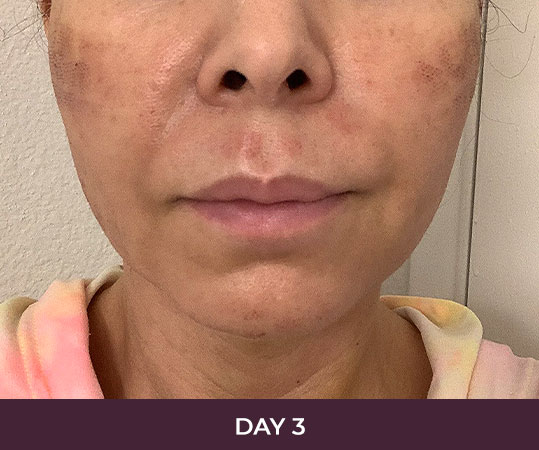
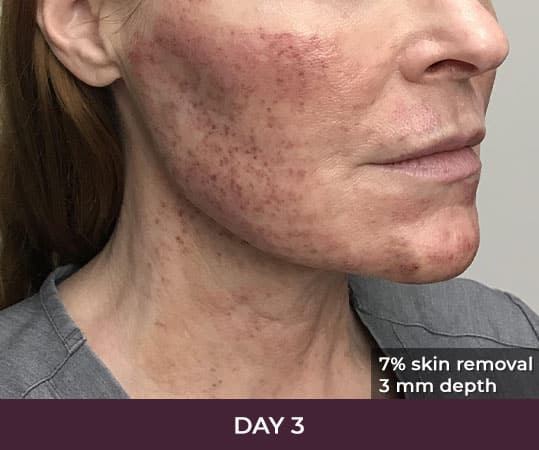
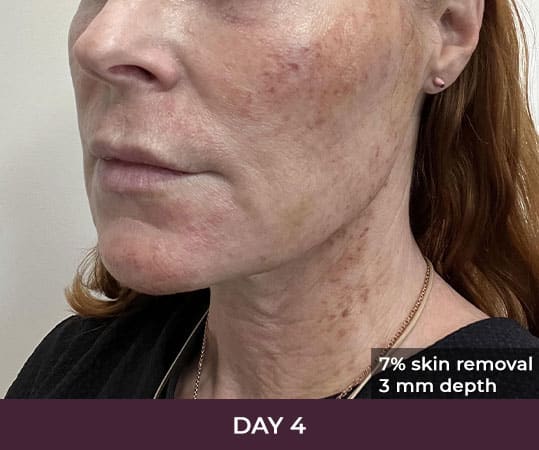
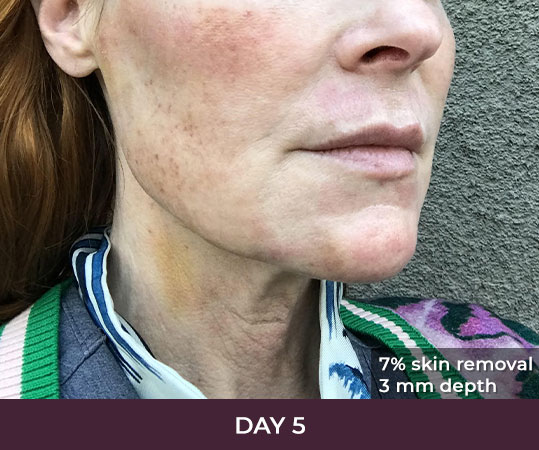
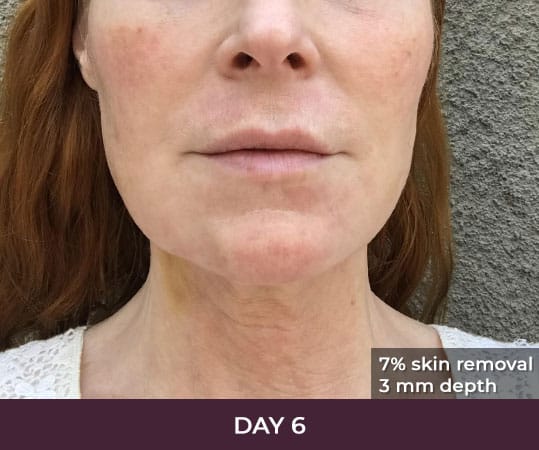
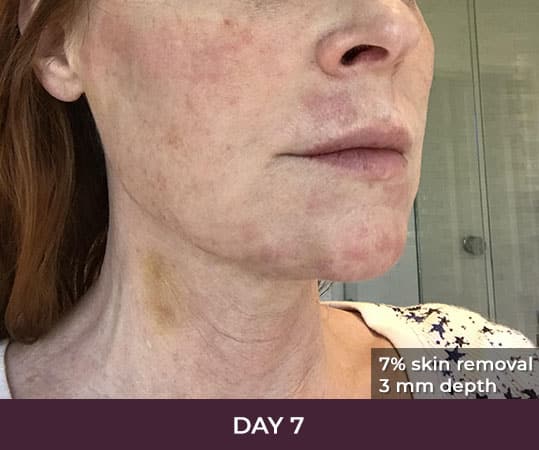
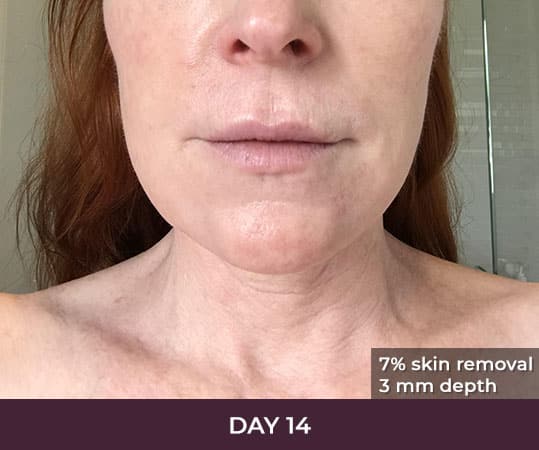
Ellacor™ Practice Before & After
Our office worked closely with Cytrellis to develop patient care protocols. As the first office in Orange County to provide Ellacor, we have treated many local and out-of-state patients since November of 2021.
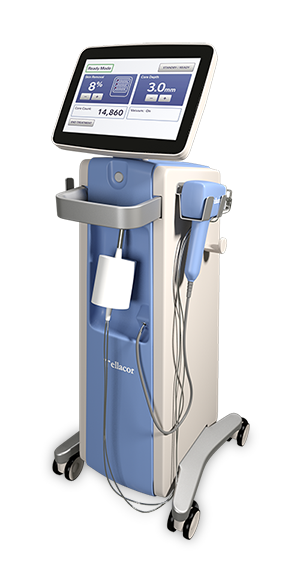
What is ellacor™ System with Micro-Coring™ Technology?
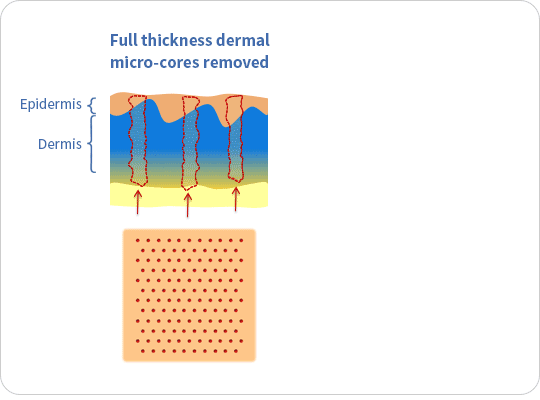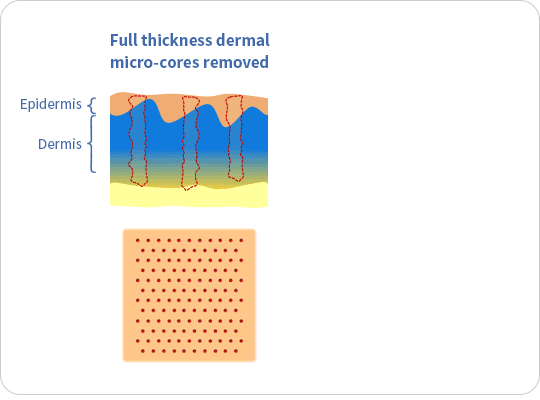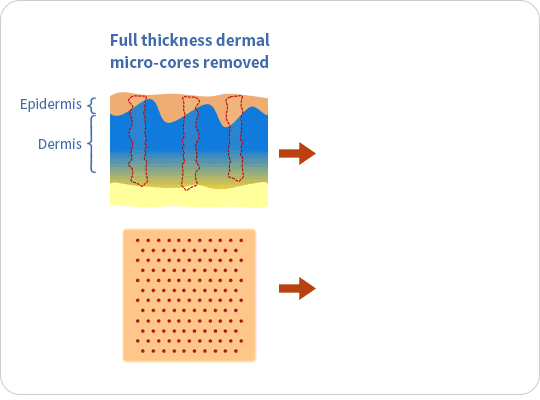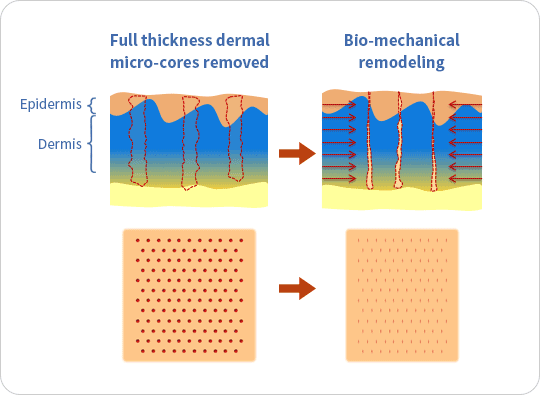
The ellacor™ System is a first-of-its-kind technology for the treatment of wrinkles that removes micro-cores of excess skin in the mid and lower face on the microscale, without surgery or the use of thermal energy.
Treatment Areas
The ellacor™ System with Micro-Coring™ Technology is indicated for use by medical professionals for the treatment of moderate to severe wrinkles in the mid to lower face.
Benefits of Ellacor™
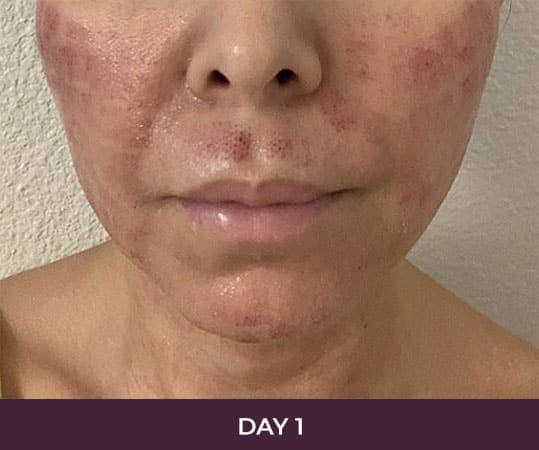
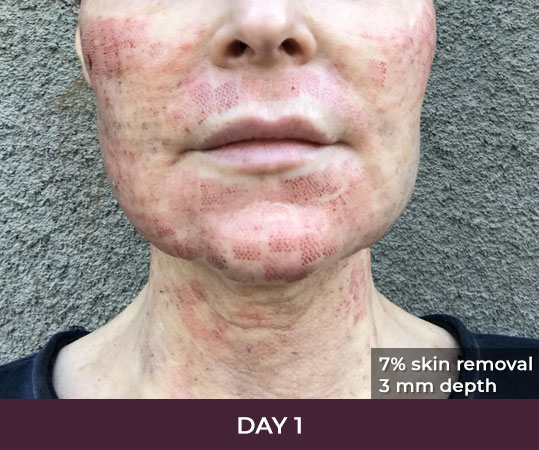
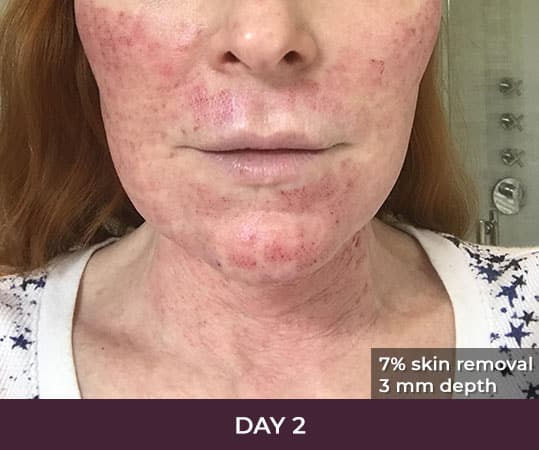
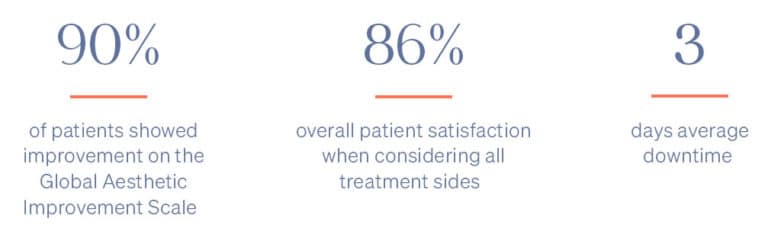
- Minimal Downtime (3-day average)
- Reduces excess skin by about 5 to 8 percent per treatment
- No Scarring
- Long-lasting Results
- No thermal injury to the skin
- A typical treatment removes equivalent of 2 square inches of skin
- Most patients in clinical trial reported pain during the procedure less than 2 on a 0-10 scale. You will provided with appropriate anesthesia prior to the procedure to make you more comfortable.
How Does Ellacor™ Work?
The ellacor™ System uses hollow needles to remove excess skin on the microscale. After the skin is removed, the body’s natural healing response goes into effect as the skin quickly realigns to close the cores.
Ellacor™ removes micro-portions of excess skin to achieve an improvement in the appearance of wrinkles without the evidence of scarring and faster healing than many energy-based procedures that use heat generated by laser, radiofrequency and ultrasound energy to induce intentional thermal injury to the skin.

Ellacor™ Reviews
See below real reviews from our practice patients. See more practice reviews at: Goole 4.9/90+ & RealSelf 4.7/190+
Treatment & Recovery
How many treatments will I need?
As every patient has different needs, we recommend you to schedule a complimentary consultation with one of our specialists to determine the right treatment plan for you.
How long is a typical treatment?
An ellacor™ treatment typically takes about 30 minutes or less. In the clinical trial, most patients underwent a series of three treatments spaced approximately 30 days apart.
Is the treatment painful?
Your provider will determine the appropriate anesthesia prior to the procedure to make you more comfortable. Most patients in our clinical trial reported pain during the procedure as less than 2 on a 0-10 scale.4
Is the recovery time long?
There is minimal recovery time following the ellacor™ procedure — 70 percent of patients in the clinical study were comfortable going in public and/or returning to work after three days or less.
Are You a Good Candidate For Ellacor™?
The ellacor™ Micro-Coring™ System is recommended for treatment of adults aged 22 years or older with Fitzpatrick skin types I-IV.
The ellacor™ system should not be used for the treatment of:
- Areas of skin with dermatosis, e.g. skin tumors, keloids or in case of predisposition to keloids, solar keratosis, warts, or birthmarks
- The area within the bony orbital rim
- Mucous membranes
- Areas where silicone or synthetic material is implanted
The ellacor™ system should not be used on the following patients*:
- Pregnant or nursing mothers
- Allergy to stainless steel
- History or presence of clinically significant bleeding disorders
- Have undergone plastic surgery of the face within the last twelve (12) months or have any facial surgical scars less than twelve (12) months old
- Have undergone injections of dermal fillers, fat, or botulinum toxin, as well as any minimally invasive/invasive skin treatment in the treatment area during the previous six (6) months
- Have scars less than six (6) months old in the treatment area
Ellacor Video Testimonials
Frequently Asked Questions
The ellacor™ System is a first-of-its-kind technology for the treatment of wrinkles that removes micro-cores of excess skin in the mid and lower face on the microscale, without surgery or the use of thermal energy.
How do I know if ellacor™ is right for me?
The ellacor™ System with Micro-Coring™ Technology is indicated for use by medical professionals for the treatment of moderate and severe wrinkles in the mid to lower face in adults 22 years or older with Fitzpatrick skin types 1-4.
How does ellacor™ work?
The ellacor™ System uses hollow needles to remove micro-cores of of excess skin, resulting in an improved appearance of wrinkles without evidence of scarring1,2 and faster healing time than many energy-based procedures.
How is the ellacor™ System different from other skin revitalization devices?
Ellacor™ removes micro-portions of excess skin to achieve an improvement in the appearance of wrinkles, whereas many other skin revitalization devices use heat generated by laser, radiofrequency and ultrasound energy to induce intentional thermal injury to the skin.
How is ellacor™ different from microneedling?
Microneedling uses small needles to injure the tissue, but it is not actually removed during this procedure – the tissue is temporarily displaced. Ellacor™ is the first device to actually remove skin on the micro-scale.
How many treatments will I need?
As every patient has different needs, we recommend you speak with your provider to determine the treatment plan that is right for you. In the clinical trial, most patients underwent a series of three treatments spaced approximately 30 days apart.
Can I receive treatment with ellacor™ while pregnant?
No. Like other aesthetic treatments, ellacor™ is not approved for use in expectant or lactating mothers.
Contraindications
The ellacor™ System with Micro-Coring™ Technology should not be used on the following patients:
- Pregnant or nursing mothers
- Suffering from open wounds, sores, or irritated skin in the treatment area
- Allergy to stainless steel
- Allergy to topical, local, or injected medications or preparations that may be used during the procedure, such as petrolatum, lidocaine, bupivacaine, chlorhexidine, or povidone-iodine
- History or presence of clinically significant bleeding disorders
- Skin or autoimmune conditions that may affect the treatment outcome; these may include, but are not limited to: actinic keratosis, raised nevi, rosacea, melasma, active acne, cutaneous papules/nodules, active inflammatory lesions, dermatitis, psoriasis, cellulitis, urticarial folliculitis, acute inflammatory phase of scleroderma, rheumatoid arthritis, eczema, allergic dermatitis, collagen disorders, or lupus
- Systemic infections or acute local skin infections (such as, Hepatitis disorders type A, B, C, D, E, or F, or HIV infection)
Treatment Precautions
Appropriateness for treatment is based on the clinical assessment of the patient by the treating physician. Use caution when treating patients with the following conditions or taking the following medications:
- History of hyperpigmentation
- Recent exposure to sun or tanning beds with re, peeling, or swollen skin
- Active, chronic, or recurrent infection including bacterial or fungal infections
- History of active herpes simplex infection in the treatment area
- Use of topical or oral preparations/medications that may change the skin integrity or prolong healing
- Over the Counter (OTC) and herbal supplements that may increase the risk of bleeding or prolong healing, such as gingko biloba, garlic, ginseng, dong quai, fever few, and fish oil
- Other medications or medical conditions that may interfere with the treatment or that the treatment provider believes mat compromise the safety of the patient or the efficacy of the treatments. Refer the patient to their Primary Care Physician (PCP) or other managing health care provider or clearance prior to treatment